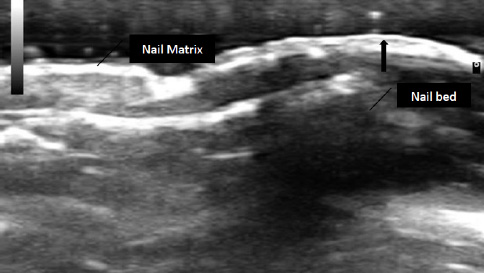
The Open Rheumatology Journal is an open-access journal that publishes research articles, reviews/mini-reviews, letters and guest-edited single-topic issues in all important areas of experimental and clinical research in rheumatology, which include epidemiology, etiology, pathogenesis, clinical outcomes and treatment of inflammatory, immunologic, pediatric and adult rheumatological and musculoskeletal conditions, as well as metabolic, and degenerative soft and hard connective tissue diseases.
The Open Rheumatology Journal, a peer-reviewed journal, is an important and reliable source of current information on developments in the field. Emphasis is placed on publishing quality papers, making them freely available to researchers worldwide.
The Open Rheumatology Journal is an international, peer-reviewed, open-access journal covering all aspects of rheumatology published continuously by Bentham Open.