All published articles of this journal are available on ScienceDirect.
Sicca Symptoms and their Association with Chronic Rhinosinusitis in a Community Sample
Abstract
Objective:
To determine associations between sicca symptoms, chronic rhinosinusitis (CRS) symptoms and asthma in a community survey.
Methods:
Data was obtained from the Spring 2009 South Australian Health Omnibus Survey which sampled, via interviewer administered questionnaire, 3007 individuals aged 15 years and over whose socio-demographic distribution corresponded to South Australian population estimates. Respondents were asked a range of questions relating to the presence of persistent dry eyes or dry mouth, CRS and medically diagnosed nasal polyps and asthma. Relationships between symptoms were explored using maximum likelihood dependency tree analysis.
Results:
The respective population prevalences were: dry mouth (5.9%), dry eyes (8.6%), nasal polyps (3.8%), CRS (13.2%) and asthma (12.0%). The overall prevalence of sicca symptoms (dry eyes or dry mouth) was 12.4%. Dependency tree analysis revealed the expected symptom clustering between (1) sicca symptoms and their association with female gender and increasing age and (2) CRS, nasal polyps and asthma (one airway hypothesis). However there was also an association between dry eyes and CRS (OR 2.5, 95% CI 1.9, 3.4), which was in fact stronger than the association between CRS and asthma (OR 1.9, 95% CI 1.4, 2.5).
Conclusions:
Sicca symptoms are common in the community. Our novel finding of a strong association between dry eyes and CRS suggests that further research into the relationship between airway inflammation and sicca symptoms is required. These findings may have particular relevance to Sjögren’s syndrome (SS) in both its primary and secondary forms.
INTRODUCTION
Dry eyes and dry mouth are collectively described as sicca symptoms. Sicca symptoms can impact substantially on a patient’s quality of life, and currently only symptomatic treatment is available, such as the use of artificial tears. The clinical relevance of sicca symptoms was demonstrated in a recent Dutch study, where over 35% of participants with sicca had consulted their general practitioner about these symptoms [1].
Isolated sicca symptoms may be associated with aging, hormonal changes, and with side effects from some pharmaceuticals. An important cause of severe sicca symptoms is Sjögren’s syndrome (SS), an autoimmune chronic inflammatory disease associated with salivary and lacrimal dysfunction [2]. Sjögren’s syndrome occurs in a primary form, pSS, which is a systemic disease associated with many extraglandular features, or it may occur secondary to other autoimmune diseases such as rheumatoid arthritis, systemic lupus erythematosus or scleroderma. Involvement of the upper and lower respiratory tract has long been known to exist in SS, and Sjögren in 1943 first described involvement of nasal glands with chronic inflammatory infiltrates [3]. An Ear Nose and Throat (ENT) review of 74 patients with pSS, rheumatoid arthritis (RA) plus secondary SS, or RA alone revealed that ENT signs and symptoms were common amongst those with sicca symptoms [4]. Pulmonary manifestations of pSS include bronchiolitis, interstitial lung disease and cystic lung disease, and bronchial hyperreactivity, which have been reported in up to 50% of patients [5-7]. A recent study reported that five patients presenting with severe chronic bronchiolitis as a feature of pSS, all had a history of recurrent sinusitis and upper respiratory tract infections [8]. Collectively, this data suggests that there may be a pathogenic relationship between sicca symptoms, and upper and lower airway inflammation, manifesting as rhinosinusitis and asthma symptoms.
The aim of this study was to determine the prevalence of sicca symptoms in an Australian population sample and to determine associations between sicca symptoms, chronic rhinosinusitis (CRS) and asthma.
METHODS
Participants
Data was obtained from the South Australian Health Omnibus Survey (SAHOS) during the Spring of 2009. The SAHOS methodology has been described in detail elsewhere [9]. Briefly, the annual survey sample is a clustered, multistage, systematic, self-weighting area sample of 4,400 households whose socio-demographic distribution correspond to South Australian population estimates. The 2009 response rate was 68%, resulting in 3007 face to face interviews with one individual per household, selected by the next birthday and aged 15 years or older.
Respondents were asked a range of health-related questions, including:
- Q1: Have you had persistent dry or gritty eyes for more than 3 months?
- Q2: Have you had a persistent dry mouth for more than 3 months?
- Q3: Have you ever been told by a doctor that you have nasal polyps?
- Q4: Have you ever suffered from sinusitis or sinus problems that lasted for more than 3 months?
- Q5: Have you ever been told by a doctor that you have asthma?
- Q6: Do you still have asthma?
Demographic data included age, gender, education level, employment status, household income, country of birth, and area of residence.
The questionnaire and methodology for this survey was approved by the North West Adelaide Health Service Human Ethics Committee.
Statistical Analysis
Population prevalence estimates and logistic regression models were analysed using Statistica v 6.1 (StatSoftInc, Tulsa, OK, USA).
Population prevalence estimates were calculated by weighting to the individual’s probability of selection according to Australian Bureau of Statistics population estimates. Asthma prevalence was determined using Q6, as this is considered the most reliable indicator of asthma status [10].
The cross-sectional relationships between variables in this study were interpreted by fitting a Chow and Liu maximum likelihood dependency tree [11] using the MIM graphical modelling software [11, 12], with the CLTree option. This analysis was performed using dichotomous variables (gender, dry mouth, dry eyes, sinusitis, nasal polyps and asthma) in a multi-way log-linear analysis, and including age as a continuous covariate.
RESULTS
South Australian population prevalence estimates of sicca (dry eyes and/or dry mouth) and CRS symptoms and asthma are shown in Table 1. The prevalence estimates for sicca symptoms, CRS and asthma were comparable (12.4%, 13.2% and 12.0% respectively), and affect a substantial proportion of the population.
South Australian Population Prevalence Estimates of Sicca and Sinusitis Symptoms and Asthma. Estimates were Calculated Using Population Weighted Data from 3007 Individuals
| Symptom | Prevalence % (95% CI) |
|---|---|
| Dry Mouth | 5.9 (5.0,6.8) |
| Dry Eyes | 8.6 (7.6,9.6) |
| Any sicca | 12.4 (11.2,13.6) |
| Chronic Rhinosinusitis (CRS) | 13.2 (12.0,14.5) |
| Nasal Polyps | 3.8 (3.2,4.6) |
| Asthma | 12.0 (10.8,13.2) |
Within sicca symptoms, dry eyes were more prevalent then dry mouth (8.6% vs 5.9%), and were observed more often in females and older age groups. Therefore the highest prevalences were observed in females over 65 years (Fig. 1). Dry mouth was associated with poorer socioeconomic status indicators and, in contrast to dry eyes, these associations remained significant after adjustment for age and gender. Multiple logistic regression analysis revealed that the lowest education level (left school at 15 years or less, adjusted OR 1.6, 95% CI 1.1,2.4, p = 0.025), and low annual income (<$20,000, adjusted OR 1.9, 95% CI 1.3,2.7, p = 0.001) were both independent predictors of dry mouth. The cross-sectional inter-relationships between gender, dry eyes, dry mouth, CRS, nasal polyps and asthma were interpreted by estimating a maximum likelihood dependency tree, which included age as a covariate. The fitted model, graphically displayed in Fig. (2), was highly significant (likelihood ratio 799.7, DF = 6, p < 0.00001), and there was no evidence of lack of fit (likelihood ratio 10.7, DF = 177, p = 1), indicating that further interactions, or higher order interactions, between variables, were not substantial.
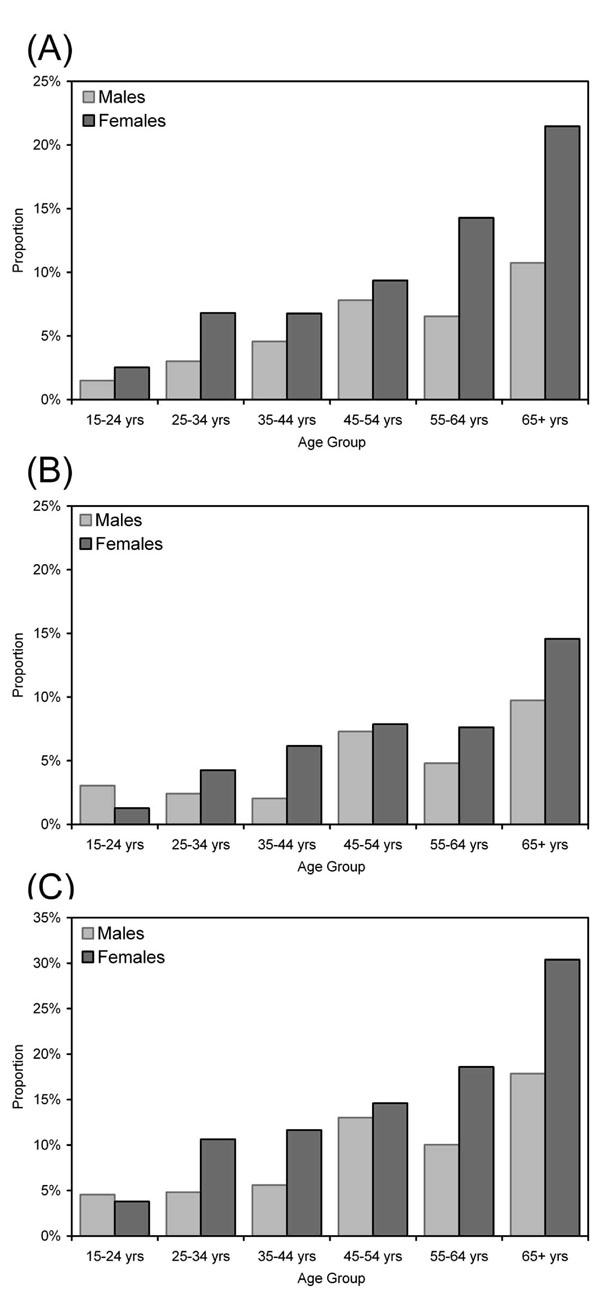
Prevalence of (A) dry eyes, (B) dry mouth or (C) either dry eyes or dry mouth (any sicca) gender and age.
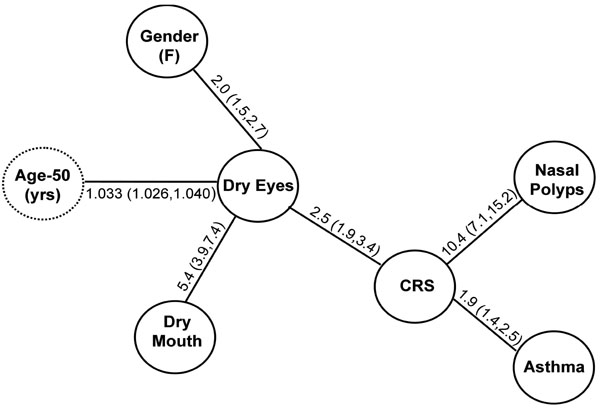
Dependency tree analysis of the relationships between gender, dry eyes, dry mouth, chronic rhinosinusitis (CRS), nasal polyps and asthma. Age (in years, centered around 50) was included as a covariate in the model. Odds Ratios (95% CI) are depicted along the edges connecting variables, and all were significant at a p-value < 0.0001. The fitted model was highly significant (likelihood ratio = 799.7, DF = 6, p < 0.0001), and there was no evidence of lack of fit (likelihood ratio = 10.7, DF = 177, p = 1) indicating that further, or higher order, interactions between variables were not substantial. The displayed model can be decomposed into 5 components: (Dry Eyes:Dry Mouth), (CRS:Asthma), (CRS:Nasal Polyps), (CRS:Dry Eyes) and (Gender:Dry Eyes/Age:Dry Eyes/Age).
The associations with dry eyes were clearly multifactorial (Fig. 2), and, as expected, there were strong associations with dry mouth, female gender and increasing age. Similarly, consistent with the “one airway” hypothesis [13], sinusitis was associated with both nasal polyps and asthma. However, there was also a novel, strong association between dry eyes and CRS (odds ratio 2.5, 95% CI 1.9, 3.4, p < 0.0001), which was marginally stronger than the previously characterised, and therefore expected, associations between dry eyes and female gender (odds ratio 2.0, 95% CI 1.5, 2.7) and between sinusitis and asthma (odds ratio 1.9, 95% CI 1.4, 2.5).
DISCUSSION
This population-based study estimates a prevalence of 5.9% for dry mouth and 8.6% for dry eyes. These estimates of sicca prevalence are lower than other population studies, whose estimates have varied widely, ranging from 11 – 30% [1, 14-17]. This may be attributable to both survey sampling and the screening questions used to assess sicca symptoms. In addition, due to the constraints of this type of community survey, we were only able to ask a single question each for dry eyes and dry mouth, and this may have reduced the prevalence estimates of these symptoms.
We were unable to perform any clinical or laboratory examinations of the participants, therefore we were unable to determine the prevalence of SS in this population. However, it is known from other studies that the prevalence of SS is much lower than that of sicca symptoms [14-17], with estimates ranging from < 1% to 3% of participants, depending on the population, age group and classification criteria used [1, 14-17].
This study demonstrates that sicca symptoms are associated with chronic rhinosinusitis (CRS) symptoms, which were both defined as symptoms of duration longer than three months. The requirement for long standing symptoms makes it unlikely that this association is due to side effects associated with over-the-counter medications used for episodic sinusitis; and the medications used for CRS are mainly topical [18]. Further, other studies have demonstrated that CRS shares a similar female predominance and increased incidence with age [19, 20] to that observed with sicca symptoms. Our observed association between CRS and sicca symptoms is a novel finding as, although it has been assumed that patients with sicca symptoms are at increased risk of CRS because of dry mucosa, this has not previously been demonstrated. This association was specifically with dry eyes, rather than dry mouth, which suggests that the association with sicca symptoms is not simply a manifestation of increased mouth breathing due to airways obstruction. Nasal polyps, which are the severe end of the spectrum of CRS, were not strongly associated with dry eyes.
We observed that CRS is a common factor in both asthma and sicca symptoms. An association between allergic rhinitis and asthma (both atopic and non-atopic) has long been noted, and has been dubbed the ‘one-airway’ or ‘united airway’ hypothesis’ [13, 21, 22]. CRS typically precedes the development of asthma, and can also contribute to unsatisfactory asthma control. Further, the presence and type of asthma is influenced by sensitization, and the duration and severity of allergic rhinitis [21]. Because our study was cross-sectional, we were unable to determine the directional nature of the association between CRS and sicca symptoms. For example, it is plausible that dry eyes may be secondary to airway inflammation associated with CRS. Equally, given that mucosal secretions contain secretory IgA and a range of effector molecules which contribute to immune defence at mucosal surfaces [23, 24], it is also plausible that mucosal dryness of the paranasal sinuses may contribute to infection susceptibility. Thickened mucosal secretions lacking the aqueous component might also contribute to poor sinus drainage and bacterial overgrowth.
One of the limitations of this study is the reliance upon self-report for a diagnosis of asthma and CRS; however it is likely that those participants who self-report current asthma, or sinusitis symptoms of more than three months duration, do actually have these conditions. Self-reported asthma has been used widely and has been shown to have good validity [10], and our prevalence estimate of 12% in the South Australian population is comparable to the national estimate of approximately 10% reported by the Australian Centre for Asthma Monitoring [25].
In conclusion, this study reports on the prevalence of sicca symptoms in a randomly selected representative population sample, and demonstrates a significant association with CRS. The relationship between CRS and both asthma and sicca symptoms, suggests that sicca symptoms, and possibly pSS, share common pathogenic mechanisms with these inflammatory disorders of the upper and lower respiratory tract. Further research into the relationship between airway inflammation and sicca symptoms is therefore warranted.
ACKNOWLEDGEMENT
This work was funded by the Hospital Research Foundation, Adelaide, Australia
CONFLICT OF INTEREST
Declared none.


