All published articles of this journal are available on ScienceDirect.
Self-Reported Symptoms in a Cohort of Rheumatoid Arthritis and Systemic Lupus Erythematosus during the COVID-19 Quarantine Period
Abstract
Background:
During the first three months of the COVID-19 pandemic in the Philippines, there was a supply shortage of hydroxychloroquine and methotrexate. Limited access to medication and the life changes resulting from the COVID-19 pandemic may predispose patients with rheumatoid arthritis (RA) or systemic lupus erythematosus (SLE) to disease flares.
Objective:
This study aimed to investigate self-reported symptoms of disease flares among patients with rheumatoid arthritis or systemic lupus erythematosus during the COVID-19 pandemic.
Methods:
A total of 512 completed online surveys from patients with SLE or RA were collected. The data included sociodemographic characteristics, self-reported physical symptoms, health service utilization, and availability of hydroxychloroquine and methotrexate.
Results:
Seventy-nine percent of respondents had lupus, while 21% had RA. One-third of the cohort had contact with their attending physician during the two-month quarantine period prior to the survey. Eighty-two percent were prescribed hydroxychloroquine and 23.4% were prescribed methotrexate; but 68.6% and 65%, respectively, had “irregular” intake of these medicines due to unavailability. The current health status was reported as good by 66.2%; 24% had no symptoms during the two-week period prior to the survey. The most common symptoms experienced were joint pain (51%), muscle pain (35%), headache (26.8%), and skin rash (19.1%). Five percent had a combination of these four most common symptoms. Irregular supply of hydroxychloroquine among patients with SLE (n=323) was associated with more frequent occurrence of muscle pain (40.6% vs 27.9%, p=0.03) or rash (27.4% vs 11.7%, p<0.001). Irregular supply of methotrexate among RA patients prescribed hydroxychloroquine and methotrexate (n=36) was associated with more frequent occurrence of joint pains with or without swelling (73.9% vs 38.5%, p=0.04). Irregular supply of hydroxychloroquine was associated with less frequent occurrence of dizziness (0 vs 66.7%, p<0.001) among RA patients (n=18).
Conclusion:
In our cohort of RA and SLE, the majority reported at least one symptom that may indicate disease flare. There was a significant association between the irregular supply of hydroxychloroquine or methotrexate with the presence of muscle pain, rash, or joint pains during the 14-day period prior to the survey.
1. INTRODUCTION
The 2019 coronavirus disease (COVID-19) pandemic has significantly affected the world for nine months and will continue to do so until an effective treatment or a vaccine is discovered. As of December 13, 2020, the infection has affected more than 70 million people and caused the death of almost 1.6 million individuals globally [1]. Containment measures that have been in place since the middle of March 2020, including community quarantine and physical distancing, have slowed down the spread of the infection. However, this has significantly limited the access of patients with chronic diseases to medical care. Among these chronic diseases are Rheumatoid Arthritis (RA) and Systemic Lupus Erythematosus (SLE) which have estimated prevalence of 0.6% and 0.03-0.05%, respectively, among Filipinos [2, 3].
During the first few months of the pandemic, several drugs, including hydroxychloroquine were evaluated in randomized clinical trials to determine their efficacy and safety in the treatment or post-exposure prophylaxis for COVID-19 [4-9]. This, and the prevailing limited mobility from 16 March to 31 May 2020, when the enhanced community quarantine was in a place affected the supply of medicines. Many patients in the Philippines with SLE and RA, among other rheumatology patients, had difficulty maintaining their supply of medications like hydroxychloroquine and methotrexate [10].
The focus on hydroxychloroquine as a possible treatment option for COVID-19 especially affected patients with SLE and RA. As an anchor drug, it is recommended for all patients with SLE at a dose not exceeding 5 mg/kg real body weight. It is effective in controlling lupus rash and arthritis and preventing SLE disease flares [11]. The 1995 HERA study showed its significant benefit on synovitis, pain, and physical disability of recent-onset rheumatoid arthritis [12]. Despite its relatively delayed onset of action, its benefits in reducing cardiovascular morbidity in patients with RA have been recognized, making it a good second-line Disease-Modifying Anti-Rheumatic Drug (DMARD) therapy for patients with inadequate response to methotrexate [13]. The surge in demand for hydroxychloroquine as a COVID treatment has severely affected access to this drug, putting additional burden on our patients with SLE and RA [14]. For Filipinos with SLE who already had increased baseline (pre-pandemic) depression and anxiety scores [15] and certain vulnerabilities, such as coping, self-esteem, and social support [16], the life changes resulting from the pandemic and quarantine, anxiety over their vulnerability to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, and lack of supply of hydroxychloroquine and/or methotrexate may predispose these patients to disease flare.
By June 2020, however, the National Institutes of Health and the World Health Organization advised against the use of anti-malarial drug based on results from randomized clinical trial in hospitalized patients that showed it has no benefit for decreasing the likelihood of death or speeding recovery among hospitalized patients with COVID-19 and in preventing the development of symptoms among exposed individuals [17, 18].
This study aimed to investigate self-reported symptoms of disease flares among patients with SLE and RA during the COVID-19 pandemic. Documentation of the health status of our patients during this health crisis will assist the government and the health sector in facilitating care for non-COVID patients whose treatment support may have been inadvertently marginalized at this time when the supplies of hydroxychloroquine and other common medications for these patients were inaccessible.
2. METHODS
2.1. Study Design
We conducted a cross-sectional online survey. A convenience sample of SLE and RA patients was recruited through their patient organizations or through their physicians who were members of the Philippine Rheumatology Association. The snowball sampling technique was implemented by sending a link to the questionnaire through various online channels (Appendix A and B).
2.2. Development of the Survey Instrument
The survey instrument was adapted from the study of Tee ML et al. [19]. A few additional questions were added to address the objectives of this study. The survey was translated from English to Filipino by the University of the Philippines Manila Sentro ng Wikang Filipino. It was field tested on ten individuals. A few revisions in terms were made to improve reading comprehension for the general public.
2.3. Patient Recruitment and Survey Administration
The e-survey was conducted from May 19 to 26, 2020, through the SurveyMonkey® platform. Patients with RA or SLE were recruited to voluntarily participate in the survey through a public invitation in social media and through their rheumatology patient groups. A link to the questionnaire was sent through social media accounts. Respondents were encouraged to share the link with as many people as possible. There was no monetary incentive offered to the participants. Access to online help links was provided. These contained information about COVID-19, availability of medicines in selected areas, and introduction to the use of teleconsultations.
The survey allowed respondents to review and change their answers through a Back button. The survey platform had an automatic method for capturing responses.
2.4. Main Outcome Variable
The survey asked for the presence of any of the following symptoms during the past 14-day period: fever, chills, headaches, myalgia, cough, difficulty breathing, chest pain, coryza, sore throat, dizziness, nausea, vomiting, diarrhea, rash, oral ulcer, joint pain with or without swelling, bubbly or foamy urine, leg or facial edema, bleeding, or convulsion.
2.5. Study Factors
We collected data on SLE and RA patients’ sociodemographic characteristics, availability of medication, and health service utilization.
2.6. Ethical Considerations
We conducted this study in accordance with the Declaration of Helsinki as approved by the UP Manila Research Ethics Board (UPMREB 2020-198-01). The introductory part of the questionnaire explained that the response to the survey implied the participant’s consent to use the collected data, with the assurance of anonymity and confidentiality in the handling of data. It explained the purpose of the study and indicated the approximate length of time to complete the survey. The survey only collected non-identifiable information.
2.7. Statistical analysis
Only completed questionnaires were analyzed. We used IBM SPSS Statistics 24.0. Percentages were calculated for sociodemographic variables, access to medicines, physical symptoms, and health service utilization variables. We used the Chi-square test of independence to test the association between the symptoms experienced and the supply of medicines (hydroxychloroquine and methotrexate) during the pandemic. However, for contingency tables where at least one assumption of Chi-square test has been violated, the Fisher exact test was used instead. The level of significance was set at 0.05.
3. RESULTS
3.1. Population Characteristics
There were 821 unique IP addresses that viewed the survey, but only 739 agreed to participate (participation rate of 90.01%). There were 521 participants who completed the survey (completion rate of 69.28%). Most of the participants were females aged 20-50 years old, had a college education, with a household size of 3-5 persons, and residents of the island of Luzon, including the National Capital Region (Table 1). Approximately, 48.2% were employed and some were health care professionals (45/512, 8.8%). Most had medical insurance (85%). The majority had a duration of illness of more than two years. There were more respondents with lupus (405/512, 79.1%) than rheumatoid arthritis (107/512, 20.9%). The rheumatoid arthritis group had a lower female: male ratio but higher proportions of individuals who were older, married, had a college education, and at least one symptom during the 14-day period prior to the survey.
Approximately, one-third of the cohort had contact or consultation with their attending physician during the two-month quarantine period prior to the survey. Ninety percent of SLE and 50% of RA were prescribed hydroxychloroquine, whereas 10% of SLE and 66% of RA were prescribed methotrexate. There were 33 SLE (8.1%) and 18 RA (16.8%) patients who were not prescribed hydroxychloroquine and methotrexate. In total, 82% had hydroxychloroquine and 23.4% had methotrexate prescriptions. However, 68.6% and 65%, respectively, had irregular intake of these medicines during the quarantine period due to unavailability.
| Variable | Diagnosed Illness | p-value | ||
|
Systemic Lupus erythematosus (N=405) |
Rheumatoid arthritis (N=107) |
|||
| Female | - | 396 (97.8) | 93 (86.9) | <0.001* |
| Age Group | - | - | - | <0.001* |
| - | 12-21 | 26 (6.4) | 0 (0.0) | - |
| - | 22-30 | 116 (28.6) | 16 (15.0) | - |
| - | 31-40 | 132 (32.6) | 24 (22.4) | - |
| - | 41-49 | 87 (21.5) | 26 (24.3) | - |
| - | 50 + | 44 (10.9) | 41 (38.3) | - |
| Education | - | - | - | 0.323 |
| - | Secondary education or less | 43 (10.6) | 15 (14.0) | - |
| - | College education or more | 362 (89.4) | 92 (86.0) | - |
| Location | - | - | - | 0.177 |
| - | NCR | 167 (41.2) | 53 (49.5) | - |
| - | Luzon | 172 (42.5) | 42 (39.3) | - |
| - | Visayas | 47 (11.6) | 6 (5.6) | - |
| - | Mindanao | 15 (3.7) | 6 (5.6) | - |
| - | Overseas | 4 (1.0) | 0 (0.0) | - |
| Household Size | - | - | - | 0.253 |
| - | 1-2 persons | 52 (12.8) | 18 (16.8) | - |
| - | 3-5 persons | 241 (59.5) | 67 (62.6) | - |
| - | 6 persons or more | 112 (27.7) | 22 (20.6) | - |
| Marital Status | - | - | - | 0.007* |
| - | Single | 202 (49.9) | 35 (32.7) | - |
| - | Married | 182 (44.9) | 64 (59.8) | - |
| - | Divorced/separated | 14 (3.5) | 3 (2.8) | - |
| - | Widowed | 7 (1.7) | 5 (4.7) | - |
| Employed | - | 193 (47.7) | 54 (50.5) | 0.605 |
| Health Care Professional | - | 35 (8.6) | 10 (9.3) | - |
| Prescribed hydroxychloroquine | - | 366 (90.4) | 54 (50.5) | <0.001* |
| - | Medications available as usual | 121 (33.1) | 11 (20.4) | - |
| - | Medications unavailable | 245 (66.9) | 43 (79.6) | - |
| Prescribed methotrexate | - | 49 (12.1) | 71 (66.4) | <0.001* |
| - | Medications available as usual | 12 (24.5) | 30 (42.3) | - |
| - | Medications unavailable | 37 (75.5) | 41 (57.7) | - |
| With at least one comorbidity | - | 244 (60.2) | 71 (66.4) | 0.248 |
| With at least one symptom | - | 313 (77.3) | 98 (91.6) | 0.001* |
3.2. Self-Reported Symptoms
The majority of respondents (345/521, 66.2%) rated their current health status as good. Twenty-four percent (25.9% of SLE and 18.7% of RA) had no symptoms. Among those symptomatic, 25% had one symptom, 19% had two symptoms, and 12% had three. Most of these symptoms were nonspecific, but some may be related to lupus or RA disease flare (joint pains, muscle pains, edema, foamy urine, rash, oral ulcer, pallor, or bleeding). Four patients tested for COVID-19 (3 SLE, 1 RA) and three (all SLE) were quarantined. However, there was no information as to when they had the tests in relation to the survey and what their symptoms were during quarantine.
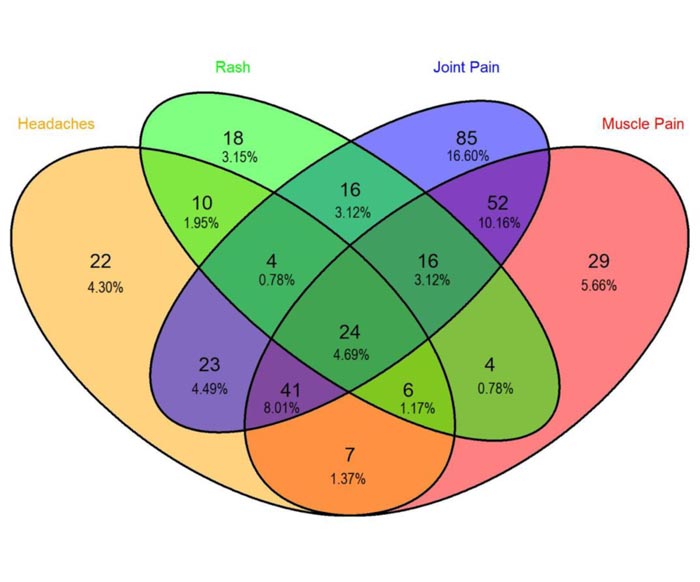
The most common symptoms during the two-week period prior to the survey were joint pain, muscle pain, headache, and skin rash, with respective percentages of 51%, 35%, 26.8%, and 19.1%, respectively (Table 2). There were combined symptoms of joint pain, headache, muscle pain, and rashes in 4.6% (Fig. 1).
Fewer than 10% of respondents had fever, chills, colds, cough, sore throat, difficulty breathing, diarrhea, bubbly or foamy urine, facial or leg edema, pallor, or bleeding.
Irregular supply of hydroxychloroquine among patients with SLE (n=323) was associated with a higher occurrence of muscle pain (40.6% vs 27.9%, p=0.03) or rash (27.4% vs 11.7%, p<0.001, (Table 3). Irregular supply of methotrexate among RA patients prescribed hydroxychloroquine and methotrexate (n=36) was associated with a higher occurrence of joint pains with or without swelling (73.9% vs 38.5%, p=0.04). Irregular supply of hydroxychloroquine was associated with a lower occurrence of dizziness (0 vs 66.7%, p<0.001) among RA patients (n=18).
| Symptoms | Diagnosed Illness |
Total (N=512) |
|
|
Systemic Lupus erythematosus (N=405) |
Rheumatoid arthritis (N=107) |
||
| Joint pain with or without swelling | 192 (47.4) | 69 (64.5) | 261 (51.0) |
| Muscle pain | 141 (34.8) | 38 (35.5) | 179 (35.0) |
| Headaches | 119 (29.4) | 18 (16.8) | 137 (26.8) |
| Rash | 86 (21.2) | 12 (11.2) | 98 (19.1) |
| Dizziness | 49 (12.1) | 10 (9.3) | 59 (11.5) |
| Cough | 33 (8.1) | 2 (1.9) | 35 (6.8) |
| Chest pain | 31 (7.7) | 6 (5.6) | 37 (7.2) |
| Bubbly or foamy urine | 27 (6.7) | 11 (10.3) | 38 (7.4) |
| Sore throat | 27 (6.7) | 2 (1.9) | 29 (5.7) |
| Oral ulcer | 24 (5.9) | 4 (3.7) | 28 (5.5) |
| Difficulty breathing | 24 (5.9) | 9 (8.4) | 33 (6.4) |
| Leg edema | 23 (5.7) | 4 (3.7) | 27 (5.3) |
| Nausea, vomiting, diarrhoea | 17 (4.2) | 3 (2.8) | 20 (3.9) |
| Facial edema | 15 (3.7) | 1 (0.9) | 16 (3.1) |
| Runny nose | 10 (2.5) | 5 (4.7) | 15 (2.9) |
| Persistent fever (>38°C for at least 1 day) | 9 (2.2) | 1 (0.9) | 10 (2.0) |
| Chills | 8 (2.0) | 3 (2.8) | 11 (2.1) |
| Bleeding | 3 (0.7) | 1 (0.9) | 4 (0.8) |
| Persistent fever and cough or difficulty breathing | 2 (0.5) | 0 (0.0) | 2 (0.4) |
| Pallor | 2 (0.5) | 0 (0.0) | 2 (0.4) |
| Diagnosis | Disease-modifying drug(s) prescribed | No. of Patients | Supply of Drug Analyzed | Symptoms | Drug Supply | ||
| Regular | Irregular | p-value | |||||
| Systemic Lupus Erythematosus | Hydroxychloroquine | 323 | Hydroxychloroquine | Muscle pain | 31 (27.9) | 86 (40.6) | 0.03 |
| Systemic Lupus Erythematosus | Hydroxychloroquine | 323 | Hydroxychloroquine | Rash | 13 (11.7) | 58 (27.4) | <0.001 |
| Rheumatoid Arthritis | Hydroxychloroquine | 18 | Hydroxychloroquine | Dizziness | 2 (66.7) |
0 (0.0) |
<0.001 |
| Rheumatoid Arthritis | Hydroxychloroquine and methotrexate | 36 | Methotrexate | Joint pain with or without swelling | 5 (38.5) |
17 (73.9) | 0.04 |
4. DISCUSSION
The benefits of hydroxychloroquine for lupus have been previously demonstrated [20-23]. Its use is recommended for all patients with SLE unless contraindicated [11]. In the survey, 90% of patients with SLE were on hydroxychloroquine. In RA, hydroxychloroquine is usually added to the treatment regimen when there is an inadequate response to methotrexate monotherapy. It is notable that 50% of our RA respondents are on hydroxychloroquine. Both hydroxychloroquine and methotrexate are slow-acting drugs with prolonged clinical effects. Hydroxychloroquine has long plasma terminal elimination half-life (approximately 40-60 days), which reflects a high volume of distribution [21]. For methotrexate, the half-life is less than 10 hours, but its active polyglutamated form may remain in tissues for extended periods and explain the prolonged effects [24].
Patients with chronic autoimmune rheumatic diseases such as SLE and RA require long-term use of combination treatments for disease control. Regular monitoring for adverse effects of drugs and episodes of disease flares are needed for careful calibration of their medications. In most instances, premature discontinuation of disease-modifying drugs leads to a flare or aggravation of disease activity [25-27]. When adequate disease control is achieved, a few studies have shown that withdrawal of these disease-modifying drugs is possible without rebound disease flare [28-30]. There are, however, limited data on the effects and safety of unplanned discontinuation of these drugs on disease activity.
Our Filipino SLE and RA cohort shared the same drug supply concern of patients in the United States, Europe and Canada [31]. In a questionnaire developed by EULAR and distributed to rheumatologists, it was concluded that there was a shortage of hydroxychloroquine, according to 49% of the respondents [32]. This is most likely attributable to the off-label use of hydroxychloroquine for treatment and prophylaxis for COVID-19, as well as the limited importation of medicines from India that adversely affected the availability of these drugs [33, 34]. Approximately, 55% of our cohort of lupus and RA resulted in abrupt, unplanned discontinuation of disease-modifying drugs. More respondents with SLE and irregular supply of hydroxychloroquine experienced muscle pain or rashes. Likewise, more RA patients receiving the combination of hydroxychloroquine and methotrexate had flares of joint pains when methotrexate supply became irregular. To begin with, these patients were likely those whose disease activity was more difficult to control.
While almost 75% reported one or more symptoms, only 37% were able to consult their attending physicians. Drug unavailability and inability to reach out to their doctors were additional challenges that they had to deal with during the quarantine period. Changes in school and job workload, financial difficulty, changing patterns of family dynamics and other human interactions were significant life events which they had to cope with as well. The feeling of helplessness may fully mediate anxiety, depression, and perceived stress among patients with SLE [35]. Daily stress and stressful life events such as these have been associated with flares of SLE [36].
In response to the situation, several medical organizations, including the Philippine Rheumatology Association, issued statements against the irrational use of hydroxychloroquine [37]. The need for rapid establishment of communication links between patients and health providers became one of the priorities of the community and the government. Our institution resorted to virtual consultation as we enhanced our telemedicine system. The Department of Health issued guidelines to enable the use of e-prescriptions sent via various tele-platforms [38]. It also set up telemedicine platforms through which people could avail of free consultations. Through these strategies, alternative therapies and other appropriate actions were advised. Counseling for apparent mental health concerns was also prioritized.
That some patients do not have the basic know-how and technical requirements for teleconsultation such as smartphones, computers, or internet limited the adoption of these innovative platforms. On-going efforts from both government and private sectors to assist these communities are expected to enhance health care delivery.
The study has several limitations. The use of an online survey has limited the subjects to those with access to the internet. There were only a few respondents from the Visayas and Mindanao where there were more reports of constraints in the supply of the medicines of concern. We do not have data on disease activity prior to the pandemic. Due to the study design, the self-reported symptoms cannot be definitely attributed to rheumatic disease flare. We did not look at factors other than supplies of hydroxychloroquine and methotrexate that may affect the occurrence of symptoms. There is no documentation of the length of time when hydroxychloroquine and/or methotrexate were unavailable. Standardized patient reported outcome measures, such as the Medical Outcomes Study Short Form-36 and Lupus Quality of Life, were not used in this study, as it was deemed difficult to deploy these measures through a survey disseminated through social media.
CONCLUSION
In our cohort of 512 patients with lupus erythematosus or rheumatoid arthritis, 75% had at least one symptom, the most common being joint pain, muscle pain, headache, and rash. Only 37% were able to access their physicians. Among those prescribed the drug, 68% and 65% had irregular supplies of hydroxychloroquine and methotrexate, respectively, during the two-month period of enhanced community quarantine in the Philippines. Irregular supply of these drugs was associated with increased occurrence of muscle pains or rash among SLE hydroxychloroquine-users and joint pains among RA patients who were taking the hydroxychloroquine-methotrexate combination. These symptoms likely indicate the occurrence of disease flares among these patients.
AUTHOR'S CONTRIBUTION
MLT, EOS, CAT, and PWR conceptualized and designed the study. EOS, HMR, GTZ and MLT acquired the data. PWR analyzed the data. All authors interpreted the data, wrote the manuscript, critically revised the manuscript for important intellectual content, and approved its final version.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the UP Manila Research Ethics Board Manila, Philippines under approval no. (UPMREB 2020-198-01).
HUMAN AND ANIMAL RIGHTS
No Animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
Written informed consent was obtained from all individual participants included in the study.
AVAILABILITY OF DATA AND MATERIAL
The data can be accessed through: https://data.mendeley. com/datasets/wss8gm4xrh/1.
FUNDING
This study did not receive any form of financial support from the pharmaceutical industry or any organization.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
We thank the Southeast Asia One Health University Network, Dr. Roger C. Ho of the Department of Psychological Medicine, Yong Loo Lin School of Medicine, National University of Singapore, Kent Ridge 119228, Singapore Professor Carol Pulumbarit of Sentro ng Wikang Filipino, the Philippine Rheumatology Association, Lupus and RA support groups in the Philippines, and the survey participants.
SUPPLEMENTARY MATERIAL
Supplementary material is available on the publishers web site along with the published article.


