All published articles of this journal are available on ScienceDirect.
Vitamin D and Type I Diabetes
Abstract
Vitamin D is a fat-soluble vitamin that plays an important role in bone metabolism but is also endowed with the capability of modulating inflammatory and immune function. Recent studies reported a relationship between low vitamin D levels and several autoimmune diseases such as Type 1 Diabetes. Vitamin D regulates the expression of over 200 genes, also related to immune modulation, suggesting a putative role in these diseases pathogenesis. This review overviews the most recent advances on the association between vitamin D and increased risk of Type 1 Diabetes as well as between vitamin D and either glucose homeostasis or insulin sensitivity. The effects of vitamin D in modulating the immune response and balancing anti-inflammatory cytokines, suggest that vitamin D system may represent an unforeseen target for the design of novel strategies for the treatment of patients with autoimmune diseases and in particular Type 1 Diabetes.
1. INTRODUCTION
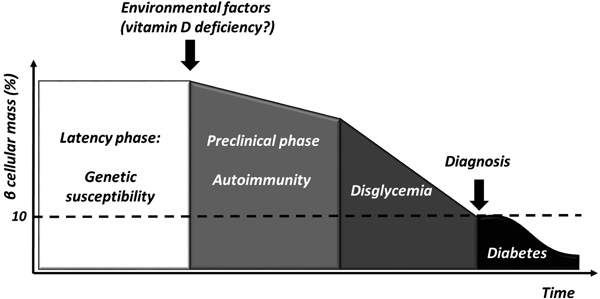
T1D is a chronic immune-mediated disease characterized by selective loss of insulin-producing pancreatic beta cells that occurs in genetically susceptible individuals [1]. However, the low concordance rate among homozygote twins [2] suggests that environmental factors might play a pivotal role in triggering beta cells destruction and clinical manifestations (Fig. 1).
Mounting evidences suggest a putative role of several agents, among which vitamin D seems to be involved. Beyond its pivotal role in regulating calcium and bone homeostasis, vitamin D exerts an action on the regulation of both innate and adaptive immunity as well as on cardiovascular and metabolic functions as VDR is nearly ubiquitously expressed in nucleated cells, in particular pancreatic beta cells [3], and in insulin responsive tissues such as skeletal muscle, myocardium and adipose tissue [4, 5].
In addition, a relationship between low vitamin D levels and the development and the severity of several autoimmune diseases such as RA, MS, SLE, IBDs, and T1D, has been reported [6-10].
This review provides a summary of the actual knowledge of the role of vitamin D in preventing and treating T1D. Since a higher prevalence of vitamin D deficiency has been reported in patients with T1D compared with the general population [11-13]. Furthermore, we discuss whether vitamin D supplementation may influence the immune regulation and the subsequent progression to T1D, in genetically susceptible individuals.
2. VITAMIN D AND IMMUNE MODULATION
A first evidence of vitamin D involvement in immune modulation derived from the isolation of the VDR in mononuclear cells. Further in vitro studies showed that 1-25(OH)2D regulates the function of almost all the main actors of immune system, acting on both innate and adaptive immunity (Fig. 2).
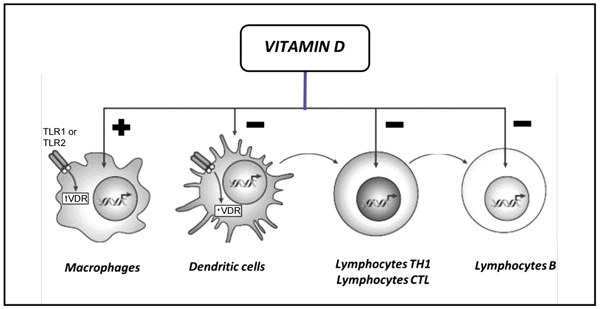
Legend: CTL, Cytotoxic; TH1, T Helper 1; TLR1, Toll-Like Receptor 1; TLR2, Toll-Like Receptor 2; VDR, Vitamin D Receptor.
2.1. Vitamin D and the Innate Immune System
Vitamin D enhances chemotaxis and phagocytic capabilities of macrophages and monocytes, which are important effector cells, fighting against pathogens. Furthermore, 1-25(OH)2D and the VDR activate the transcription of antimicrobial peptides such as defensin β2 and cathelicidin antimicrobial peptide via toll-like receptor signaling and the production of other cytokines such as INF-γ and IL-4 [14, 15] so 1-25(OH)2D is essential for effective pathogen clearance. In the later stages of infection, 1-25(OH)2D plays a role in the contraction of the immune response and has an anti-inflammatory effect on macrophages characterized by a decreased production of pro-inflammatory factors and an increased production of anti-inflammatory, promoting the macrophages M2 phenotype while inhibiting the M1 phenotype and thereby restoring the balance between these subsets [10, 16].
Moreover, monocytes and other innate APCs, in particular DCs, are important targets for the immunemodulatory effects of vitamin D. DCs are crucial in initiating effective adaptive immune responses against pathogens, and for maintaining self-tolerance and immune homeostasis. 1-25(OH)2D induces an immature-like tolerogenic state in DCs characterized by decreased production of pro-inflammatory factors like IL-12 and TNFα and increased anti-inflammatory IL-10 production [17-19]. These tolerogenic DCs are less able to promote the proliferation and cytokine production of pro-inflammatory T cells, while they enhance the differentiation of Treg cells and induce the apoptosis of autoreactive T cells [17-19]. Interestingly, a positive autocrine loop exists in DCs and other immune cells, which allows to activate locally vitamin D because these cells express the enzyme CYP27B1 [20]. This expression leads to achieve a high local concentration of the active form of vitamin D, required for immunomodulatory effects.
2.2. Vitamin D and the Adaptive Immune System
Early studies investigating the effects of vitamin D on adaptive immune system demonstrated the expression of the VDR and vitamin D-activating enzymes in both T- and B cells. In B cells, 1-25(OH)2D inhibits the proliferation and induces apoptosis; furthermore, it leads to the inhibition of plasma cells and the differentiation of post-switch memory B cells, and significantly reduces immunoglobulin secretion [21].
The other major type of adaptive immune cells, T cells, is also an important target for the immunomodulatory effect of vitamin D. In this context it influences cell proliferation and differentiation, and modulates their cytokine production leading to a shift from a proinflammatory to a more tolerogenic immune status. 1-25(OH)2D acts as an inhibitor of T cells cytotoxic activity, by suppressing Fas-Ligand expression in activated T cells [22]. Moreover, it drives CD4+ differentiation suppressing Th1 and Th17 functions and enhancing a more favorable and less inflammatory Th2 or Treg phenotype [23, 24].
Despite the consistent body of evidence provided by in vitro studies, the role of vitamin D in the development and treatment of autoimmune conditions is still largely debated and in vivo studies are less conclusive.
3. VITAMIN D AND BETA CELL FUNCTION
Beyond its immunomodulatory and anti-inflammatory action, Vitamin D also promotes pancreatic beta cell function via different mechanisms. Firstly, it exerts a direct action enhancing insulin secretion and beta cell survival by inactivation of NF-κb and other cytokines and exerting an anti-apoptotic effect via the down regulation of Fas-Ligand [25]. Furthermore, vitamin D could indirectly affect insulin secretion by regulating intracellular calcium. In fact, insulin secretion is a calcium-dependent process and is influenced by calcium flux through the cell membrane. Vitamin D regulates calbindin, a cytosolic calcium-binding protein found in beta cells [25]. Moreover, vitamin D influences intracellular calcium even in insulin responsive tissues, such as skeletal muscle and adipose tissue, promoting insulin sensitivity. At this level, vitamin D enhances insulin sensitivity also by directly stimulating the expression of insulin receptors and/or by activating peroxisome proliferator-activated receptor-δ [26]. Finally, vitamin D inhibits rennin formation and local rennin-angiotensin-aldosterone system that impairs the action of insulin in vascular and skeletal muscle tissue leading to altered glucose uptake and insulin resistance [27].
4. VITAMIN D AND AUTOIMMUNE DISEASES
Autoimmune diseases are the result of a loss of immune homeostasis and an altered self-antigen recognition leading to the destruction of body tissues by autoreactive immune cells. The pathogenesis of these diseases is multifactorial and involves a combination of genetic predisposition, epidemiological risk factors and environmental contributors. One important factor may be the availability of sufficient vitamin D levels. In this context, in vivo studies with animal models of various autoimmune diseases showed that supplementation with 1-25(OH)2D could prevent the initiation and the progression of experimental models of MS and RA [28, 29], decreases the incidence of diabetes and the severity of IBDs and SLE [30, 31]. From the other side, VDR and vitamin D deficiency have been correlated with a worsening of arthritis and IBDs severity [32].
In humans, the fist body of evidences derives from the inverse relationship between UV exposure and the prevalence of some autoimmune diseases like T1D, IBDs, and MS [33-35]. Furthermore, the incidence of RA and T1D decreases after vitamin D supplementation [36-38]. Moreover, a normal vitamin D status is paramount in preventing RA related osteoporosis and the correction of a deficient vitamin D status should be suggested to each rheumatic patient [39, 40]. However, investigating the relationship between vitamin D intake and the prevalence of autoimmunity is challenging because dietary intake and UV exposure are estimated. Therefore, many studies focused on the correlation between serum 25(OH)D levels and autoimmune diseases. In fact, patients with autoimmune diseases, like MS, RA, T1D, and IBDs had lower 25(OH)D levels compared with controls and higher disease activity [7, 13, 41, 42]. Furthermore, some VDR polymorphisms have been associated with the risk of developing autoimmune diseases [43]. In conclusion, autoimmunity has been related with vitamin D deficiency, intake, UV exposure, and VDR polymorphisms and, in experimental models, 1-25(OH)2D supplementation shows promising results. Although human studies are still inconclusive, these data warrant further investigation to establish if at-risk subjects could benefit from vitamin D treatment and more randomized-controlled trials are still needed.
5. VITAMIN D AND GENETIC PREDISPOSITION TO T1D
Besides the well-established association with HLA genes, more than 40 non-HLA genes, putatively linked to T1D, have been identified through a whole genome SNPs genotyping approach, most of them involved in vitamin D functions and immune regulation [44]. In particular, alterations or polymorphisms in the genes coding for VDBP, VDR or enzymes involved in 1-25(OH)2D activation have been studied in T1D individuals.
Conflicting results come from the association studies between polymorphisms in the genes involved in vitamin D function and T1D. In fact, an association between VDR polymorphisms and T1D, has been reported in Chilean, German and Taiwanese [45-47], but not in Portuguese and Finnish populations [48, 49]. More recently, a meta-analysis reported an association between the SNPs BsmI and Taq in the VDR gene and the susceptibility to T1D in children [50, 51]. Consistently, a systematic review by Tizaoui et al. suggested that VDR polymorphisms are involved in T1D pathogenesis [52].
An association between low levels of VDBP and T1D has been described by Blanton et al. [53], although it is still unclear whether it could be related to VDBP polymorphisms [30-33]. By contrast, studies on the relationship between CYP2R1 and CYP27B1, two hydroxylase involved in vitamin D pathway, and T1D susceptibility, led to conflicting results in different ethnic groups [54-58].
A recent meta-analysis concluded that several genes of the vitamin D system show a significant association with T1D [43], highlighting a predisposing role for VDR polymorphisms and for the CYP27B1 gene, which encodes the main enzyme for vitamin D activation. In addition, Norris et al. have recently reported the association between plasma 25(OH)D concentrations and islet IA in children at increased genetic risk of T1D. Such an association was modified by VDR rs7975232, as increased 25(OH)D was related to reduced IA risk [59].
6. VITAMIN D AND RISK OF T1D
A birth-cohort study in 10366 northern Finnish infants who received cholecalciferol supplementation since one year of age, revealed, irrespective of dose, and regardless baseline 25(OH)D status, a reduced risk of developing T1D throughout adulthood. Furthermore, in children who received at least the recommended vitamin D dose (2000 units/day) regularly, this risk was reduced by about 80% [60].
A systematic review and meta-analysis [61] of four case-control studies and one cohort study concluded that vitamin D supplementation could significantly reduce the risk of developing T1D, as vitamin D supplemented children had a 29% reduction in the risk of developing T1D compared to their unsupplemented peers. Moreover, there was some evidence of a dose-response effect, with those using higher amounts showing a lower risk of developing T1D and a suggestion that also the timing of supplementation might influence the subsequent risk. Such a reduced risk was also revealed in a cohort of patients with early diagnosis of rickets who were prone to develop T1D [60].
A recent meta-analysis of studies with a case control or cohort design, aimed at evaluating the correlation between the risk of developing T1D to the intake of vitamin D either in early life or during pregnancy. A significant inverse association between early vitamin D intake and the risk of T1D was found in 5 out of 8 studies [38], whereas the association between maternal intake of vitamin D and risk of T1D in the offsprings is still controversial [62]. Notably, Talaat et al showed that higher levels of islet antibodies, anti-glutamic acid decarboxylase antibodies and anti-insulin antibodies were significantly associated with 25(OH)D deficiency, supporting a role of vitamin D in the pathogenesis of T1D [63] and, as previously discussed, increased 25(OH)D levels have been correlated with a reduction of IA risk. In particular, in the TEDDY nested case-control study, being vitamin D sufficient (> 50 nmol/l) during infancy and childhood was associated with a reduction of risk of IA of 40% and 31%, respectively compared with those that were insufficient [59].
In our previous studies, we found, in our region, a frequent vitamin D insufficiency in pregnancy and in newborns especially among migrants [64, 65]. Moreover, we highlighted an association between vitamin D status at birth and risk of having type 1 diabetes up to 10 years of age, also if only in the subgroup of migrant babies. Vitamin D levels were significantly lower in migrant than in Italian newborns and values lower than < 2.14 ng/ml had a high or to developing T1D [66, 67].
Taken together, the results of the available studies showed that vitamin D deficiency is associated with an increased risk of IA and of developing T1DM later in life, at least in some selected populations and that vitamin D supplementation could reduce this risk with a dose and timing-dependent effect.
7. VITAMIN D AND BLOOD GLUCOSE CONTROL IN T1D
The achievement of an adequate blood glucose control in patients with T1D is pivotal to avoid long-term micro and macro vascular complications.
Over the years scientific literature focused on the possible role of vitamin D supplementation on metabolic status, in particular as a possible tool to get a better blood glucose control and a reduced insulin requirement.
A retrospective study by Nwosu et al. which included T1D and T2D patients, reported a statistically significant decrease in HbA1c values following either ergocalciferol and cholecalciferol supplementation in T2D but not in T1D subjects [68]. Moreover, the relationship between 25(OH)D and HbA1c was not confirmed in a recent multivariate model analysis of 7-18 years-old children with T1D [69].
In a pilot study, Perchard et al. analyzed the effects of oral supplementation with cholecalciferol in two cohorts of T1D children, either white Caucasians or south Asians with serum 25(OH)D levels < 50 nmol/l. In spite of an inverse relationship between HbA1c and 25(OH)D levels in Caucasian, but not in Asian patients, the treatment failed to influence HbA1c [70]. Conflicting results emerged from a recent retrospective study aimed at evaluating the effects of cholecalciferol on blood glucose control. This study demonstrated that both vitamin D and HbA1c pre-treatment levels highly influenced the effects of the treatment, insofar that the higher HbA1c and the lower vitamin D pre-treatment levels, the greater the reduction in HbA1c (p<0.001) after treatment with cholecalciferol can be [71]. In addition, a cross sectional Italian study confirmed the widespread hypovitaminosis D in children with T1D and reported lower 25(OH)D levels in migrants as compared to the Italian population. Notably, children with 25(OH)D levels < 25 nmol/l had at onset more severe ketoacidosis and a greater insulin requirement at discharge. Moreover, 25(OH)D status was negatively related to HbA1c and daily insulin dose during a 6 to 12 month follow-up [13].
Altogether, recent reports from literature confirmed that lower serum vitamin D levels are detected in children with T1D and significantly associated with higher levels of HbA1c and impaired metabolic control [13, 63, 70, 71]. Interestingly, vitamin D supplementation can potentially improve blood glucose control [13, 63, 70, 71] suggesting a pivotal role in regulating insulin sensitivity and glucose homeostasis (Table 1).
| Study | Participants | Vitamin D Supplementation | Mean 25OHD Levels After Supplementation |
25OHD >75 nmol/l (% subjects) |
Association 25OHD/HbA1c | Association Supplementation/HbA1c | Results |
|---|---|---|---|---|---|---|---|
| Savastio et al. [13] | 141 T1D (35 migrants, 106 Italians) | Subjects with 25OHD <75 nmol/l Cholecalciferol 1000 IU/day |
56.6±23.7 nmol/l | 20.6% | + | + | At baseline: significant inverse correlation between 25OHD levels and diabetic keto-acidosis severity. Relationship between HbA1c and 25OHD levels in a multivariate regression analysis. |
| Talaat et al. [63] | 250 T1D and 250 healthy controls | None | / | / | + | Not evaluated | Significant negative relationship between 25OHD levels and HbA1c values. Levels of islet antibodies, anti-GAD antibodies and anti-insulin antibodies were significantly associated with 25OHD deficiency. Significant association between 25OHD deficiency and high levels of pro-inflammatory cytokines. |
| Nwosu et al. [68] | Total population: 88 T1D, 43 T2D Subjects with 25OHD <50 nmol/l: 33 T1D, 31 T2D |
Subjects with 25OHD <50 nmol/l Ergocalciferol or cholecalciferol 400-7.000 IU/day for 8-16 weeks |
T1D: 67.6±20.6 nmol/l T2D: 60.7±27.7 nmol/l |
Not reported | Not evaluated | - | No changes in total daily dose of insulin per kg body weight in T1D and T2D. Significant decrease in HbA1c in T2D after 3 months of vitamin D supplementation but not in T1D. |
| Al Sawah et al. [69] | 197 T1D | None | / | / | - | Not evaluated | No differences in HbA1c across 25OHD levels (≤50; 50-75; >75 nmol/l). Not significant correlation between 25OHD and HbA1c in the multivariate model. |
| Perchard et al. [70] | Total population: 51 T1D (35 Caucasian, 11 South Asian, 5 other) Subjects with 25OHD <50 nmol/l: 40 T1D |
Subjects with 25OHD <50 nmol/l Cholecalciferol 100.000 IU (2-10 years)-160.000 IU (>10 years) single dose |
Not reported | Not reported | Only in white Caucasian subgroup | - | At baseline: no significant correlation between 25OHD and HbA1c in the whole cohort. Significant inverse relationship between 25OHD and HbA1c in the Caucasian group. After treatment: no difference in HbA1c at 3 months or at 1 year. |
| Giri et al. [71] | Total population: 271 T1D Subjects with 25OHD <50 nmol/l: 124 T1D |
Subjects with 25OHD <50 nmol/l Cholecalciferol 400 IU (25OHD 30-50 nmol/l)-6.000 IU (25OHD <30 nmol/l)/day for 3 months |
65.3±9.3 nmol/l | Not reported | + | + | Significant positive association between baseline HbA1c and HbA1c reduction after treatment. Significant direct relationship between baseline 25OHD levels and HbA1c reduction after treatment. |
CONCLUSION
Vitamin D may play a role in either T1D pathogenesis and insulin sensitivity. Although conflicting data on the effects of vitamin D on blood glucose control have been reported in literature, vitamin D supplementation should be considered a promising tool for the prevention of T1D. Indeed, most factors may account for such a reported variability, as study design, sample size, dosage, type and duration of vitamin D supplementation, sampling season and other confounding variables (such as honeymoon, anthropometric factors, etc). Randomized controlled trials with long follow-up periods are required to confirm the pathogenic role of vitamin D in T1D and to define the best formulation, the optimal dose and the appropriate timing and duration of supplementation.
Notably, mounting evidences emphasize a possible role of vitamin D in the regulation of the immune response but plasmatic 25(OH)D concentrations required to exert an immunomodulatory role in T1D have not yet been established. Furthermore, it seems that an early vitamin D supplementation, during the first year of life, might reduce the later risk of T1D but the best time to supplement children with a current diagnosis of T1D is still unknown although it appears reasonable to precociously correct vitamin D deficiency to have an immunomodulatory effect on IA and a positive action on glucose metabolism. This might envisage new perspectives in the design of targeted therapies aimed at preventing and even controlling T1D.
LIST OF ABBREVIATION
| APCs | = Antigen Presenting Cells |
| DCs | = Dendritic Cells |
| HbA1c | = Glycated Hemoglobin |
| IA | = Islet Autoimmunity |
| IBDs | = Inflammatory Bowel Diseases |
| MS | = Multiple Sclerosis |
| RA | = Rheumatoid Arthritis |
| SLE | = Systemic Lupus Erythematosus |
| SNPs | = Single Nucleotide Polymorphisms |
| T1D | = Type 1 Diabetes |
| T2D | = Type 2 Diabetes |
| Treg | = regulatory T cells |
| VDBP | = Vitamin D Binding Protein |
| VDR | = Vitamin D Receptor |
| 1-25(OH)2D | = 1,25-dihydroxyvitamin D |
| 25(OH)D | = 25-hydroxyvitamin D |
CONSENT FOR PUBLICATION
Not applicable.
CONFLICT OF INTEREST
All the authors approved the final version of the manuscript that has not been previously published anywhere and has not been under consideration in any other journal. All the authors report no conflicts of interest.
ACKNOWLEDGEMENTS
Author Contributions: SS, FC and GB conceived and designed the review; SS, GG, SB, AG, and GB contributed to the articles selection. SS, SB, AG. FC, MB and LM wrote the manuscript.


