All published articles of this journal are available on ScienceDirect.
Vitamin D, Inflammation and Immunity: Review of Literature and Considerations on Recent Translational and Clinical Research Developments
Abstract
The most relevant and recent literature findings linking exposure to sunlight, Vitamin D (VD), inflammation and immune system in health and disease, are reviewed.
Reduced sunlight exposure determined hypo-vitaminosis D to be common among patients or even healthy subjects, especially at higher latitudes. Numerous studies support the hypothesis that VD insufficiency could contribute to the higher autoimmune diseases incidence in the same geographic areas.
In the present review, the ways in which VD was reported to influence immune system, contributing to organism homeostasis or disease development are addressed. In fact, some of the hormone activities were recognised to determine stimulation or inhibition of immune system components.
Several diseases, where an association with VD deficiency was studied, are summarised. Finally, the rationale for optimization of substitutive/additive therapy with VD analogues and the last innovations regarding these drugs are mentioned.
1. INTRODUCTION
Vitamin D3 (VD3) is a steroidal hormone, synthesized mainly in the skin from 7-dehydrocholesterol through ultraviolet B (UVB) radiation, and in smaller percentage provided by the diet. Further metabolic passages include conversion in the liver to 25-hydroxyVD3 (25(OH)D3), its circulating form, and activation to 1,25-dihydroxyVD3 (1,25(OH)2D3) by the 1-α-hydroxylase enzyme, in kidney proximal tubule cells [1].
The metabolite measured in blood to establish VD levels is 25(OH)D, being representative of the organism VD storage [2]. The usual target range for 25(OH)D is 30-60 ng/mL or 75-150 nmol/L [3].
As reported before, cutaneous synthesis provides 80%-100% of VD requirements, but it is to be mentioned that the natural solar source for starting the metabolic pathway is not equally distributed all over the world [4]. Environmental factors associated with changing solar zenith angle, such as season, latitude, time of day, determine UVB radiation strength and its consequent capability of stimulating VD synthesis. In fact, UVB reaches to the earth surface only when the sun is sufficiently high to penetrate all atmosphere layers. This situation verifies only in the middle of the day and, beyond the 40th parallel in mid and high latitude regions, only for a few months, from the middle of spring until the end of summer. Therefore, almost in all Europe and a large portion of North America and of the former Soviet Union countries, exposure to sun includes UVB only during this short time period [5].
Relevantly, those are the same regions where the highest prevalence of autoimmune diseases is observed.
Likewise, other steroidal hormones, VD3 appear to influence immune responses, in addition to its well-known effect on calcium and bone metabolism [6, 7].
Studies performed in the last twenty years of the past century, on patients with granulomatous diseases, such as sarcoidosis, in which high VD serum levels are present, showed expression of the 25OHD-1-α-hydroxylase enzyme in granulomas forming macrophages [8]. In fact, conversion of 25-OHD to 1,25(OH)D occurs in immune system cells, expressing a different form of 1-α-hydroxylase enzyme and capable of converting the precursor to the active form. The extra-renal enzyme is not regulated by parathyroid hormone, but seems to be induced by cytokines, such as interferon (IFN)-γ, interleukin (IL)-1, or Tumour Necrosis Factor (TNF)-α [9]. Lately, in sarcoidosis, presence of a high number of T regulatory cells (Tregs) was demonstrated, together with the peripheral energy of immune system cells [10].
Interestingly, 1-α-hydroxylase enzyme forms were also described in keratinocytes and consequently, they were considered as a source for autocrine synthesis of 1,25(OH)2D [11].
A pleiotropic role for VD3 is supported also by the presence of VD Receptor (VDR) on various cell types, in healthy and diseased tissues like bone marrow, brain, colon, breast, malignant and immune cells [12]. In immune system cells, VDR is present on monocytes and macrophages, dendritic cells, T cells and B cells. Consequently, it was postulated that VD can have, in the periphery, a paracrine or autocrine action, binding on responsive elements (VDRE), located on DNA, and changing the way immune system genes are expressed [13].
Several studies testified the correlation between acquired VD deficiency and reduced immune function or autoimmunity development, cancer and even cardiovascular diseases [14-18].
Also, the potential for confounding factors was investigated, since the presence of a systemic inflammatory response was reported to be associated with lower levels of micronutrients like albumin or lipid soluble vitamins, in plasma [19].
1.1. Vd and Innate Immunity
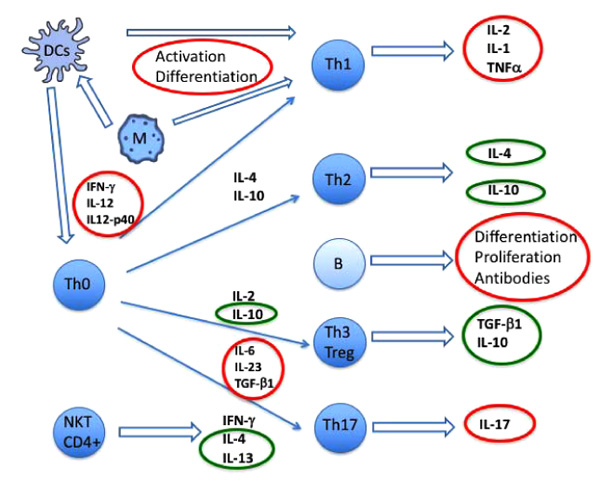
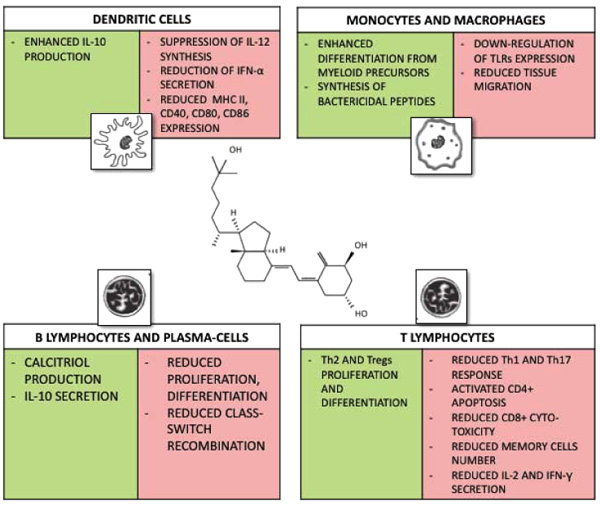
VD has both stimulatory and inhibitory effects on innate immune cells. Macrophages, monocyte-derived dendritic cells (DCs) and dermal DCs own the whole enzyme supply needed to convert VD3 into 1,25(OH)2VD3 Figs. (1 and 2) [20, 21].
According to in vitro studies, VD could contribute in the induction of differentiation of immature myeloid cells into monocytes and macrophages [22]. Moreover, VD3 deficiency and hypo-functional VDR polymorphisms are involved in susceptibility to Mycobacterium tuberculosis and Mycobacterium leprae [23]. Interestingly, studies on Mycobacterium tuberculosis resistance showed that an important role is exercised by VD in the innate immune system. In fact, VD seems to be implicated in toll-like receptors (TLRs) dependent pathways, leading to synthesis of bactericidal peptides like cathelicidin and beta defensin 4, in circulating monocytes and tissue macrophages. Binding of macrophages TLR2 or TLR4 with microbial peptides determines hyper-expression of 1-α-hydroxylase and VDR. The result is the formation of a heterodimer by 1,25OHD-VDR and retinoid X receptor (RXR) that is able to act over VDRE on cathelicidin and beta defensin 4 genes, enhancing their transcription. The cited mechanism is determinant for the killing of intracellular bacteria [24]. Moreover, induction of superoxide production by THP-1 cells infected with live Mycobacterium tuberculosis and treated with VD3 was described [15].
Other studies postulated a VD3 mediated downregulation of TLRs expression on monocytes, of TLRs mediated innate immune responses and consequently also a limitation of T helper (Th)1 activity [25].
VD is able to suppress the synthesis of IL-12, reduce IFN-α secretion and increase the production of IL-10 by DCs. Nutritional supplements containing VD determine higher IL-10 blood levels [26, 27]. IL-10 is known to down-regulate inflammatory responses reducing antigen presentation by DCs and macrophages [28]. DCs are also inhibited in their differentiation, maturation and immuno-stimulatory function following MHC class II molecules and CD40, CD80 and CD86 reduced expression [29] (Figs. 1 and 2).
A regulatory function was also reported for VD on leukocyte migration, explained possibly by a reduced peripheral chemokine synthesis: decreased synthesis of CCL2, CCL3, CXCL10 in Experimental Autoimmune Encephalomyelitis (EAE) and of CCL2, CCL5, CXCL10 in non-obese diabetic (NOD) mice [30, 31].
1.2. Vd and Adaptive Immunity
Activated T and B cells possess only the enzymatic activity for converting 25(OH)D3 to 1,25(OH)2D3 [32].
VD was correlated with reduced lymphocytes Th1 and Th17 responses and increased Th2 and Tregs activation [33]. Inhibitory effects are especially exercised on memory T cells, in concordance with their higher VDR expression [34]. In vitro, VD3 inhibits T cell proliferation, IL-2 and IFN-γ secretion and CD8 T cell cytotoxicity [35, 36].
The decrease IL-2 and IFNγ production is mediated by binding of the VDR–rXr complex to promoters of genes encoding IL-2 and IFNγ [37, 38]. The effect on Th17 cells was attributed to inhibition of IL-6 and IL-23 production [39].
CD8+ T cell deficiency characterizes many autoimmune diseases. An interesting hypothesis on autoimmune diseases development was proposed in 2012. The Authors postulated that VD lower levels at higher latitudes could facilitate the development of auto-reactive B cells, by aggravating a deficient control of Epstein Barr virus infection from CD8 T cells [40].
VD has also inhibitory effects on B cells and plasma-cells, reducing their proliferation, differentiation and Ig secretion. In B cells, VDR receptor activated signal cascade leads to impaired class switch recombination, with following reduced IgE synthesis. Up-regulation of IL-10 production in DCs can contribute to this process, inhibiting IL-4 mediated IgE synthesis [41]. Interestingly, it was demonstrated that cultures of activated B lymphocytes produce calcitriol and express IL-10 in response to autocrine and exogenous calcitriol production (Figs. 1 and 2). The relevant immuno-modulatory role of B cells, through production of IL-10, was demonstrated in ulcerative colitis treated with anti-CD20 (Rituximab). Mucosal B cell reduction and following fall of local IL-10 levels led to higher disease activity after treatment [42]. Expression of 1-α hydroxylase in B cells seems to be dependent on T cell help, through stimulation with CD-40 and IL-4 [43].
Also, VD substitutive therapy is reported to reduce Th1 and Th17 and increase Tregs responses. Consequently, also an indirect effect on differentiation, proliferation and antibody production could be postulated for B cells during treatment with VD analogues [30, 44].
2. HYPOVITAMINOSIS D AND AUTOIMMUNE DISEASES
From the clinical point of view, hypo-vitaminosis D was associated with higher risk of auto-immune rheumatic diseases, especially for Systemic Lupus Erythematosus (SLE), Rheumatoid Arthritis (RA), Systemic Sclerosis (SSc), Idiopathic Inflammatory Myopathies (IIM), but also Multiple Sclerosis (MS), Inflammatory Bowel Diseases (IBD), type 1 diabetes and other autoimmune endocrine diseases [45-51].
Interestingly, VD deficiency was associated with an increased autoimmune response also in healthy individuals. In these subjects, it was demonstrated that VD deficiency was associated with increased auto-immune response with Anti-Nuclear Antibody (ANA) positivity and higher interferton (IFN)-α activity [43].
It is to be cited that also VDR mutations with reduced function were reported in several autoimmune diseases [26, 52].
Many studies on animal models attest the usefulness of supplementation with VDR agonists in prevention and treatment of arthritis, autoimmune diabetes, EAE, IBDs, psoriasis and SLE [53-59].
Interestingly, Sainaghi et al. demonstrated, in a large retrospective study, on patients affected by autoimmune rheumatic diseases, a greater trend to development of higher parathyroid hormone (PTH) responses, compared to unaffected subjects with similarly low VD levels. In the study, the Authors observed that VD plasmatic concentrations, age and an autoimmune rheumatic disease, were independent predictors for hyperparathyroidism. Therefore, a hypothesis for an impairment of VD metabolism in course of inflammation has been developed, characterized by a scarce suppressive activity of VD on PTH secretion. Consequently, the research concludes that VD concentrations should always be assessed together with that of PTH, calcium and phosphate, to evaluate VD metabolism correctly. Moreover, the Authors postulate that VD supplementation should, therefore, be targeted to obtain PTH suppression [60]
A following study from the same group, demonstrated in autoimmune rheumatic diseases affected patients, that a treatment with a single oral dose of cholecalciferol 300,000 IU, followed by oral cholecalciferol 800-1000 IU daily for 6 months, was a more effective regime for determining PTH suppression [61].
2.1. Systemic Lupus Erythematosus
Many clinical studies focused on the relation between hypo-vitaminosis D and SLE [62]. SLE patients are by definition VD deficient, because instructed on sun avoidance and use of sunscreens [43, 63].
Some authors studied the seasonality of disease flares, showing that SLE reactivation was correlated with lower VD serum levels, in wintertime [64].
A meta-analysis of eleven heterogeneous articles showed in most of the papers an inverse correlation between serum VD levels and SLE disease activity [65].
Regarding organ specific aspects, an association was found by several studies between VD levels, general cardiovascular risk, vascular stiffness and endothelial dysfunction, in SLE patients [66, 67]. Moreover, hypo-vitaminosis D was also associated with SLE with cognitive impairment, fatigue and sleep disorders [68, 69].
In SLE patients, several prospective studies (each with a different treatment schedule) were performed on VD supplementation, with contrasting results. Terrier et al. described an increase in naive T CD4+ and regulatory T cells and a decrease in Th1, Th17 and B memory cells numbers [70].
Andreoli et al. described higher levels of Tregs cells and Th2 cytokines after long term monthly treatment with cholecalciferol [71].
An intensive regimen of VD supplementation in SLE patients with inactive disease, showed to obtain sufficient levels of serum 25(OH)D, but no effect on disease activity or serology [72].
On the other hand, Aranow et al. described failure of a VD supplementation, up to 4,000 IU daily, in diminishing the IFN signature in VD-deficient SLE patients in a 12-week double-blind, placebo-controlled trial. The Authors hypothesized that higher VD levels sustained for a longer period may be required to affect immunologic outcomes [73].
Polymorphisms of VDR seem to account for the individual response to VD, and it was found that some are associated with increased risk of SLE development and to disease phenotype [74, 75].
2.2. Rheumatoid Arthritis
Multicentre studies have shown significantly lower 25(OH)D serum levels in RA patients compared to matched controls [76]. It was recently demonstrated that VD serum concentrations appear to correlate with clinimetric indexes for quality of life, disease activity and disability in a cohort of RA European patients [77, 78]. The Authors constructed a new Patient Reported Outcome (PRO) questionnaire for RA patients’ self-estimation of the risk for VD insufficiency/deficiency-related clinical implications (D-PRO) [70].
Many studies have linked a higher risk of RA development to higher latitudes especially in winter months [79]. Lower UV-B light exposure was extensively studied as a risk factor for RA development in prospective cohort studies, the Nurses’ Health Study (NHS) and the NHSII, confirming a significantly decreased risk with higher UVB exposure especially in the absence of sun-protective behaviours [80].
It was also demonstrated in RA that winter or spring season of symptom onset can act as an independent predictive factor for joint structural damage progression at 6 months and radiographic progression at 12 months [81].
Presence of severe 25(OH)D deficiency (<10 ng/ml) was associated with worsening of symptoms like musculoskeletal pain in RA, while improvements are seen after supplementation [82, 83]. Pre-treatment of human macrophages with oestrogens can reproduce the hormonal milieu of the inflamed RA joints, where aromatase is activated and pro-inflammatory cytokines are released. It was demonstrated that VD treatment blocks completely oestrogen related aromatase activation [84, 85].
2.3. Systemic Sclerosis
In SSc patients, low VD serum concentrations have been shown [86]. Arnson et al. demonstrated, in a large multinational SSc patients’ population, that lower serum VD concentrations were inversely correlated with the extent of cutaneous fibrosis [87].
A correlation with severe nailfold video-capillaroscopic alterations and worse quality of life parameters were reported in patients with diffuse SSc form and with lower 25(OH)D serum concentrations [88].
Moreover, in a large study on SSc patients from two European reference centres, serum 25(OH)D deficiency was found to correlate with severe lung involvement, peripheral vascular, kidney and gastrointestinal Medsger’s disease severity scale parameters. Furthermore, 25(OH)D serum levels were reported to correlate with seasonality, while a supplementation with standard doses of oral cholecalciferol was found not effective in normalising serum 25(OH)D values [89, 90].
2.4. Idiopathic Inflammatory Myopathies
VD deficiency was reported in patients with Idiopathic Inflammatory Myopathies (IIM) like Polymyositis (PM), Dermatomyositis (DM), Inclusion Body Myositis (IBM) compared to controls matched for age, gender and month of serum sampling. VD levels were found to be lower in patients more close to diagnosis than in those with established disease, suggesting that low levels of VD may constitute a risk factor for developing IIM. As for RA, the IIM cases were found to be diagnosed more frequently during winter or spring. Finally, higher serum anti-Jo-1 antibodies title were shown to be correlated with lower serum 25(OH)D levels [49].
2.5. Multiple Sclerosis
Multiple studies strongly suggested that an insufficient exposure to sun or VD assumption, during the first part of life, constitutes a major risk for MS development [91, 92].
Supplementation with VD has shown to have multiple positive immunological effects in patients affected by MS. Higher Tregs count, IL-10 cytokine activity and reduction of the pro-inflammatory Th17 lymphocytes, of IL-17 and of B-cell immunoreactivity were achieved [93-95]. Additionally, low serum 25 (OH)D and a recent Epstein-Barr Virus (EBV) infection were found to be associated with blood donors with the first clinical MS manifestation [96-97].
3. VD SUBSTITUTIVE/ADDITIVE THERAPY
A recent consensus conference confirmed the necessity of VD and calcium supplementation in patients on therapy with steroids or other drugs that may reduce bone mineral density. 25OHD levels are considered to be measured once a year and replaced in those patients with insufficient/deficient levels [98, 99].
A Cochrane Review in 2014 stated that VD3 seemed to decrease mortality in elderly people living independently or in institutional care, while VD2, alfacalcidol and calcitriol had no statistically significant beneficial effects on mortality [100].
The existence of a synergism between VD3 and glucocorticoids was demonstrated: when VD3 is assumed combined with dexamethasone, there seems to be significant additive effects on inhibition of lymphocyte proliferation and Th1 cytokine production [101].
Moreover, it was demonstrated, even in healthy subjects, that VD supplementation (140,000 IU at baseline and after 4 weeks) seems to determine significant increase of Tregs percentage, suggesting a possible use of VD as additional therapy in autoimmune diseases [102, 103].
Different results of in vitro and animal models studies on VD supplementation, compared to studies performed on humans, may be justified by the fact that, while in the first two cases the active form of the molecule is used, in the second most of the times the precursor activity is evaluated. This difference determines the existence of a phase of the study where the quantitative and qualitative characteristics of the molecule activity are poorly controllable. Part of such variability may be due to the individual VDR receptor polymorphisms and/or to epigenetic changes.
However, peripheral conversion of pro-VD in calcitriol sustain the use of the first as a therapeutic agent, being a storage molecule with longer half-life and reduced hypercalcaemic effect [16].
A link between inflammation and neoplastic transformation is increasingly supported by recent literature and VD anti-proliferative actions suggested its possible use also in different types of cancers [104, 105]. VD3 showed anti-melanoma activity in experimental models, and defects in VD signalling seemed to contribute to tumour progression and disease outcome [106]. Studies on animal models of breast cancer have shown several tumour suppressive actions of VDR agonists, while human VD deficiency is common in breast cancer patients and low VD status was reported to enhance the risk for neoplasms development or progression [107]. VD/VDR deficiency was reported also in prostate cancer patients together with higher C reactive protein and IL-8 levels, providing evidence for an association among prostate cancer, VD deficiency and inflammation [108].
CONCLUSION
In conclusion, from available literature it can be observed clearly that VD is a determinant factor for immunomodulation. It has to be said that inflammatory reactions could ‘consume’ VD in autoimmune diseases, and hypovitaminosis could be a consequence. However, this would not be coherent with the results of genetic studies specifically linking genotypes regulating VD metabolism to higher autoimmunity susceptibility and with the findings of the numerous epidemiological studies cited [109]. Moreover, the beneficial effects of VD supplementation, observed in several conditions, further testify for a major role of VD in immuno-modulation [110].
The increasing interest for VD and VDR in bone biology, metabolic diseases, immunological disorders and cancer, took pharmaceutical companies to develop over more than 3,000 VD analogues in the last 30 years.
Next studies will probably continue to concentrate on new direct VDR agonists with a better biological profile for applications in therapy. Knowledge of structures of the VDR ligand-binding domain and its ligands will lead to the creation of VDR agonists with increased affinity for VDR receptor and augmented molecular metabolic stability.
As a consequence, a higher preventive and therapeutic potential, also for extra-skeletal chronic diseases, such as autoimmune and cardiovascular diseases, cancer, diabetes and infections, will be obtained.
DCs= dendritic cells; Th= T CD4+ helper cell subsets (Th1, Th2, Th3-Treg, Th17) originating from naïve T cell (Th0). Thin arrows (left) indicate cytokines that induce differentiation of Th0 cells and thicker arrows (right) indicate cytokines produced by activated Th cell subsets. B cells and Natural Killer T cells (NKT) are also reported.
Red circles indicate cytokines/activities inhibited by VD while green circles indicate cytokines enhanced by VD.
CONSENT FOR PUBLICATION
Not applicable.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


