All published articles of this journal are available on ScienceDirect.
Hematological Manifestations in Pediatric Systemic Lupus Erythematosus (SLE) Patients and Their Association with Disease Activity
Abstract
Introduction
Hematological involvement is frequent in pediatric SLE patients, and common manifestations include anemia, leukopenia, lymphopenia, and thrombocytopenia. An association has been observed between various hematological parameters and pSLE disease activity. The study aims to compare the hematological manifestations in pSLE patients with healthy controls and evaluate the association of various hematological parameters with SLE disease activity.
Methods
This cross-sectional study was conducted at the Pediatric Rheumatology Division, Department of Pediatrics, Bangabandhu Sheikh Mujib Medical University, Dhaka, Bangladesh. This study included thirty newly diagnosed pSLE patients who met the ACR revised classification criteria and thirty healthy controls. Detailed history, physical examinations, baseline investigations, reticulocyte count, and Coombs’ test were recorded. Disease activity was assessed by using the SLE Disease Activity Index (SLEDAI).
Result
All the pSLE cases (100.0%) had anemia followed by leukopenia, thrombocytopenia, and lymphopenia. Compared to controls, pSLE cases had significantly lower hemoglobin level, total white cell count, and platelet count. Hematological involvement was present in 100% of cases, followed by constitutional symptoms, mucocutaneous, musculoskeletal, renal, and neurologic involvement. Autoimmune hemolytic anemia was found in 10% of cases in this cohort. The majority (63.3%) of patients had severe disease activity (SLEDAI: >12). There is a significant negative correlation between hemoglobin (Hb) levels and SLEDAI scores in pSLE patients.
Discussion
Hematological involvement was the most common finding in this cohort, which included anemia, leukopenia, thrombocytopenia, and lymphopenia. These frequencies align with some studies (e.g., KSA thrombocytopenia) but differ from some others (e.g., KSA leukopenia 30%). Fifty percent had a positive Coombs test, consistent with a Bangladeshi study, although lower than the Turkish (76.6%) study. True AIHA was found in 10%, similar to Brazilian (14%) and other Bangladeshi studies (10-16%). pSLE patients had significantly lower Hemoglobin (Hb), Total white blood cell, and Platelet counts compared to healthy controls, consistent with previous literature. The majority of pSLE patients (63.3%) had severe disease activity, with a mean SLEDAI score of 17.1±6.75, similar to an Indonesian study but lower than the mean score in an Egyptian study. Significant negative correlation was found between hemoglobin level and SLEDAI score in pSLE patients, but no significant correlation was found between white blood cell count, platelet count, and SLEDAI score, which agrees with the studies of Samohvalov, Ahmed, and Shamim but does not match the findings of Maher. The reason behind the discrepancies in hematological parameter associations across studies is the wide spectrum of presentation, non-specific symptoms, and the multifactorial origin of cytopenias.
Conclusion
Anemia was the most common hematological abnormality in this cohort. Autoimmune hemolytic anemia was found in 10% of pSLE patients, and hemoglobin level showed a negative association with SLEDAI. Hematological parameters of pSLE patients were significantly reduced compared to healthy controls.
1. INTRODUCTION
Systemic lupus erythematosus (SLE) is a chronic, autoimmune disorder that involves various organs and systems of the body [1]. Pediatric SLE (pSLE) is more severe and aggressive with widespread organ involvement and a higher SLEDAI score at onset compared to adults [2, 3]. Among Pediatric rheumatic diseases, around 10% have been suffering from pSLE, as found in a tertiary hospital study conducted in Bangladesh [4].
Hematological involvement is very common in pSLE patients. Antibody-induced damage of blood cells by complement-dependent or independent mechanisms has been considered a pathogenic mechanism for cytopenia in SLE. Autoantibody-mediated damage to erythrocytes, suppression of erythropoiesis, blood loss, and some drugs (NSAIDs, steroids, cyclophosphamide, MMF) may be the cause of anemia. [5]
According to the American College of Rheumatology (ACR) hematological disorders criteria, hemolytic anemia, leukopenia (< 4,000 leukocytes/cumm), lymphopenia (< 1,500 lymphocytes/cumm), and thrombocytopenia (<100,000 thrombocytes/cumm) are the hematological manifestations of SLE [1].
Reticulocyte count is a good indicator for distinguishing the causes of anemia. Increased numbers of reticulocytes are found in the hemolytic process [5]. A Brazilian study reported that approximately 14% of pSLE patients had autoimmune hemolytic anemia.[6] Coomb’s test was positive in 52.3% of patients with pSLE in a Bangladeshi study [7].
Leukopenia is common in SLE and reflects disease activity, while neutropenia can result from an immune mechanism or bone marrow dysfunction. In a study by Janoudi, leukopenia was noted in approximately 50% of patients [5]. Thrombocytopenia is due to immune-mediated destruction of thrombocytes. Thrombocytopenia has also been reported in many SLE patients [8].
The Systemic Lupus Erythematosus Disease Activity Index (SLEDAI) was developed and validated as a clinical index for measuring disease activity and is widely used to assess disease activity in SLE [9]. Disease activity index showed different types of relationships with various hematological parameters [10]. Different studies also showed significant differences between the hematological parameters of pSLE patients and healthy controls [11]. Hematological abnormalities are common in pSLE, but their association with disease activity is different and remains understudied in Bangladesh. This study aimed to evaluate the hematological manifestations in pSLE patients and their association with SLE disease activity. The study also compared hematological parameters in pSLE patients with those of healthy controls.
2. METHODS
This cross-sectional analytical study was conducted at the Pediatric Rheumatology division, Department of Pediatrics, Bangabandhu Sheikh Mujib Medical University (BSMMU), Dhaka, Bangladesh, from November 2021 to June 2023. Purposive sampling was done. Thirty newly diagnosed pSLE cases fulfilling the ACR criteria available at that period, both in outpatient and inpatient departments, and age- and sex-matched thirty healthy controls were included in this study. Informed written consent was obtained from parents, and ethical clearance was also taken from the Institutional Review Board of BSMMU (NO. BSMMU/2021/8748) before enrollment.
Children with other hematological disorders causing hemolysis, e.g., Lymphoma, and Leukemia, who had a recent history of blood transfusion (within 4 months) and were taking any hematinics, and children with any other chronic disease (Chronic Kidney disease, Chronic liver disease, Tuberculosis, Thalalassaemia, or Aplastic anemia) were excluded from this study. SLE cases were diagnosed and managed by the attending pediatric rheumatologist.
Data were collected in a pretested questionnaire that included demographic information, detailed history, and physical examination. Age, sex, SLEDAI, CBC, Coomb’s test, and reticulocyte count were used as variables. Laboratory investigations included complete blood count, urine routine examination, serum ALT, serum creatinine, ANA (IF method), Anti-ds-DNA, serum C3, serum C4, 24-hour UTP, Coomb’s test, and Reticulocyte count.
Disease activity was evaluated at diagnosis by using the SLE disease activity index (SLEDAI). The total SLEDAI score falls between 0 and 105, with higher scores representing higher disease activity [12].
Autoimmune hemolytic anemia in a pSLE was defined as the presence of anemia, Coomb’s test positivity, and reticulocytosis [6].
Data were checked, verified, and analyzed by using SPSS version 26. Numeric data were expressed as mean ± standard deviation, and qualitative data were expressed as frequency and percentage. Unpaired t-test, Chi-square test, and Pearson correlation test were done to analyze the data, and a p-value < 0.05 was considered statistically significant for all tests.
3. RESULTS
The mean age of cases was 11.78±3, and 80% were female. The mean age of controls was 11.78±2.97 with female predominance (73.3%). There were no significant differences. In 50% of SLE patients, the disease duration was 6-12 months. All the pSLE cases (100.0%) had anemia followed by leukopenia, thrombocytopenia, and lymphopenia in 13.3%, 10.0%, and 20.0% of the patients, respectively, and Coomb’s test positivity was present in 50% of the cases (Table 1).
| Hematological Manifestation | Number of Patients | Percentage |
|---|---|---|
| Anemia | 30 | 100.0 |
| Leukopenia | 4 | 13.3 |
| Thrombocytopenia | 3 | 10 |
| Lymphopenia | 6 | 20.0 |
| Coombs test positive | 15 | 50 |
Compared to controls, pSLE cases had significantly lower hemoglobin levels, total count, and platelet levels. Neutrophil, lymphocyte, and reticulocyte counts did not show any significant differences between the groups (Table 2).
| Variables | Case (n=30) | Control (n=30) | p-value |
|---|---|---|---|
| Hemoglobin (gm/dL) | 8.63±1.35 | 11.97±1.37 | <0.001 |
| Total WBC counts | 5876.7±2727.9 | 9221.3±2663.2 | <0.001 |
| Neutrophil (%) | 61.53±17.22 | 56.93±6.46 | 0.325 |
| Lymphocyte (%) | 33.57±17.00 | 33.20±6.52 | 0.936 |
| Platelet (μL) | 264833.3±143096.8 | 357000.0±124639.5 | 0.040 |
| Reticulocyte count (%) | 1.07±1.82 | 1.60±0.39 | 0.271 |
Hematological involvement was present in 100% of cases, followed by constitutional symptoms (99%), mucocutaneous (96.7%), musculoskeletal (50%), renal (53.3%), and neurologic involvement (40%) (Fig. 1: Distribution of clinical manifestations among pSLE patients). Autoimmune hemolytic anemia was found in 10% of cases in this cohort (Fig. 2: Types of anemia). The majority (63.3%) of patients had severe disease activity (SLEDAI: >12), followed by moderate disease activity (SLEDAI: 6-≥12), and the mean SLEDAI score was 17.1±6.75 (Table 3).
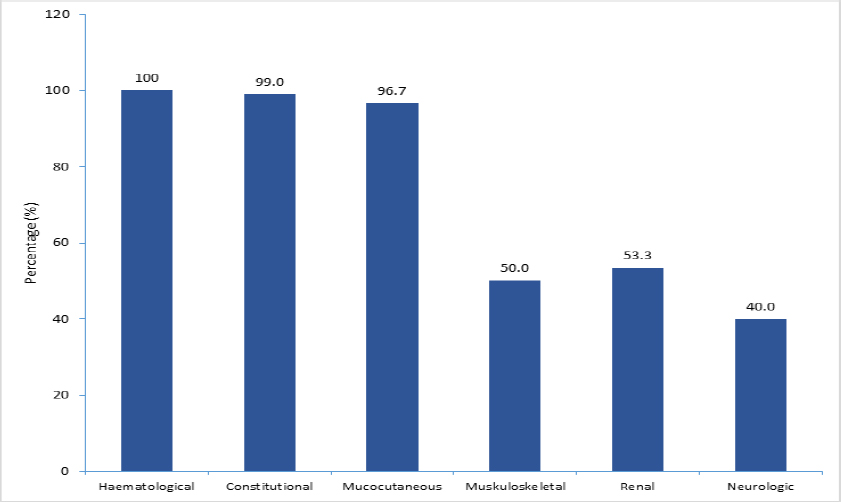
Clinical manifestations of pSLE patients.
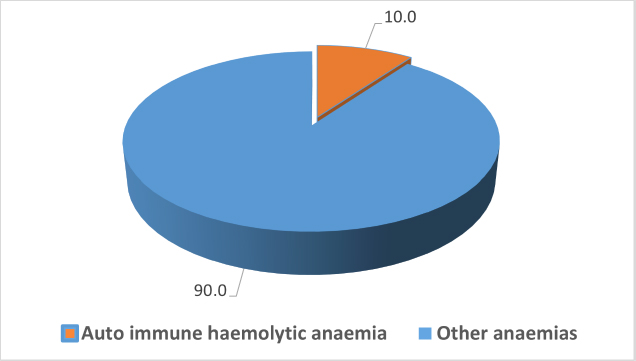
Types of anemia in pSLE patients.
| Severity of Disease | Number of Patients | Percentage |
|---|---|---|
| Moderate (SLEDAI: 6-≥12) | 11 | 36.7 |
| Severe (SLEDAI: >12) | 19 | 63.3 |
| Total | 30 | 100.0 |
| Mean±SD | 17.1±6.75 |
There is a significant negative correlation between hemoglobin (Hb) levels (Y-axis) and SLEDAI scores (X-axis) in pSLE patients. (r=-0.950, p<0.001). (Fig. 3: Relation between Hb and SLEDAI).
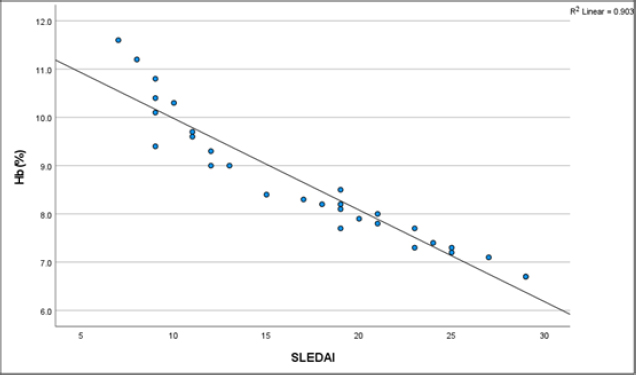
Relationship of Hb with SLEDAI in pSLE patients.
There was a weak negative correlation between white blood cell (WBC) count, platelet count, and SLEDAI scores in pSLE patients, which is not statistically significant (Figs. 4 and 5: Relation of WBC and Platelet with SLEDAI).
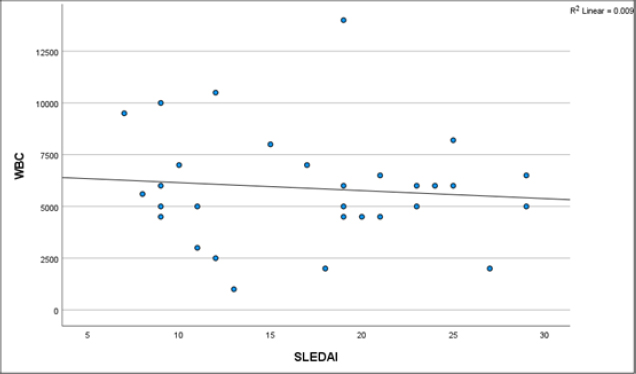
Relationship of WBC with SLEDAI in pSLE Patients.
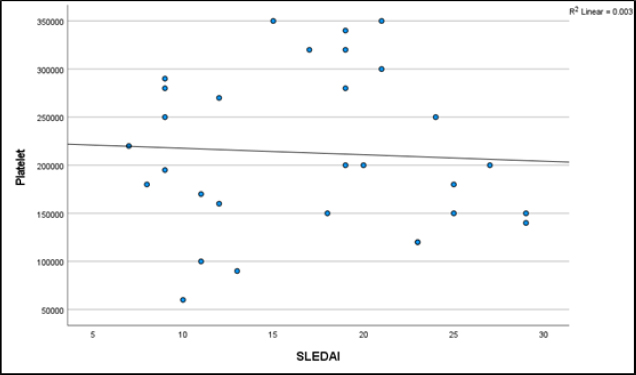
Relationship of platelet count with SLEDAI in pSLE patients.
4. DISCUSSION
Pediatric systemic lupus erythematosus (pSLE) is a multi-system autoimmune disease. Hematological involvement is common and presents a wide range of diversity [13]. Common hematological findings in affected individuals include Anemia, leukopenia, lymphopenia, and thrombocytopenia. [14].
In this study, the mean age of the pSLE patients was 11.78 years, while in the control group, it was 11.73 years. There is no significant difference, consistent with two Bangladeshi studies [7, 15] where the reported mean age was 11.5 years and 11.7 years, respectively. A similar age range (11 to 12 years) was found in a study conducted in the USA [16].
Most of the patients were female in both the case and control groups, accounting for 80% and 73.3%, respectively, which aligns with a study in China where 83.1% of pSLE patients were female [17]. The F: M ratio of this study was 4:1, consistent with a study where the F: M ratio was also 4:1, and this ratio has changed to 17:1 in the adult group [18]. In another study in the USA, the ratio had changed from 3:1 to 9: 1 when they reached puberty [19]. Hormonal effect, such as Estrogen, is thought to be the reason for this [20].
Hematological involvements are frequently observed findings among pSLE cases. A study conducted in KSA reported 63% had anemia, 30% had leukopenia, and 10% had thrombocytopenia [21]. These findings show some variation with the present study, where 100% of patients had anemia, 20% lymphopenia, 13.3% leucopenia, and 10% had thrombocytopenia. This agrees with two Bangladeshi studies. [7, 8]. An Indian study by Hasan M. et al. supports these findings, where they found that 100% patients presented with anemia.[22] But it differs from a study in Latin America, where only half of the patients (50%) had anemia. [23]. This may be due to the nutritional influence of the affected individuals.
The difference in various hematological parameters between pSLE cases and healthy controls is observed in different studies [11, 24]. Significant differences in the hemoglobin level, total WBC count, and platelet level between pediatric SLE patients and the control group (p < 0.001) were observed in the present study as well.
A study conducted in Bangladesh at the same center revealed that 92.3% of pSLE patients had constitutional manifestations, 87.7% mucocutaneous manifestations, and 52.3% had neurological manifestations [7]. The present cohort had similar findings. Another study reported that the majority of patients (92.8%) presented with constitutional symptoms, followed by mucocutaneous, renal, vascular, and serositis symptoms as observed in an Egyptian study [3]. There were variations in presentation among the above-mentioned studies, which could be attributed to geographical, ethnic, and sample size differences.
In pediatric Systemic Lupus Erythematosus (pSLE), autoimmune hemolytic anemia is antibody-mediated, often resulting in a positive direct Coombs test [1]. A study in Turkey by Gokce et al. reported that 76.6% of patients tested positive for the Coombs’ test [25]. Our study revealed 50% of patients with a positive Coombs test, similar to a study in Bangladesh where 52.2% of patients tested positive [7]. Autoimmune hemolytic anemia (AIHA) in SLE is mainly due to antibody-induced erythrocyte damage by complement-dependent or independent mechanisms [1]. In our study, 10% of pSLE patients had AIHA, a finding similar to that of a Brazilian study, which reported AIHA in 14% of patients [6]. Two Bangladeshi studies by Rahman SA. et al. and Chanda UK. et al. reported 16% and 10% of patients presented with AIHA, respectively, in their study [14, 15], which aligns with the present study.
In this study, other types of anemia were found in 90% of cases. In another study in Bangladesh, they found 55% of patients presented with anemia of other types [14]. An Indian study found that most patients had anemia due to chronic disease, whereas only 2-3% presented with autoimmune hemolytic anemia [26].
Systemic Lupus Erythematosus Disease Activity Index (SLEDAI) has been used as a validated index of a global measure of disease activity in SLE [9].
In the present study, 36.7% and 63.3% of patients were found to have moderate and severe disease activity, respectively. In an Indonesian study, severe and moderate disease activity was present in a total of 90% of patients [27]. The Mean SLE disease activity score of this study was 17.1±6.75 in the current study. A study in Egypt reported that the mean disease activity of their patients was much higher (27±19.968) [3].
Several studies have shown a variable relationship between hematological parameters in pSLE cases and disease activity. In the present study, hematological parameters showed a different relationship with disease activity. There was a significant negative relation between Hb level and disease activity, but no significant relation between total count, platelet count, and disease activity. The findings of our study are similar to the results of Samohvalov et al. where patients with low hemoglobin levels had higher disease activity (p 0.018) [10, 28]. In other studies by Ahmed H. et al. and Hakan K. et al. it was found that the SLE disease active group had a lower mean hemoglobin level [29, 30]. However, the study by Maher et al. did not find any significant relationship between Hb and SLEDAI [13]. In a study in KSA, they did not find any significant relationship between leucocyte and platelet count with disease activity, which matches our study result. [31]. Another Study by Shamim R. et al. also supports our findings, where they found a negative correlation between Hb and SLEDAI, but no significant relationship between SLEDAI and WBC and Platelet count [32].
These differences may be explained by the fact that the presentation of pSLE ranges from relatively mild illness to severe, life-threatening manifestations. Headaches, oral ulcers, arthralgia, fever, and weight loss are nonspecific and occur in children for various reasons. Also, these hematological manifestations may be due to the disease itself, another concomitant disease, or iatrogenic [13].
5. STRENGTHS AND LIMITATIONS
This study supports that Hb level is a valuable marker for the disease activity of pSLE patients, measured by SLEDAI. Besides, the cross-sectional design demonstrates association but cannot establish causal relationships between hematological parameters and disease activity. The relatively small sample size from a single center limits the statistical power for subgroup analyses and may affect the generalizability of the findings. Furthermore, the focus on newly diagnosed patients may introduce selection bias, as this cohort likely represents a more severe disease spectrum. Finally, potential confounding factors such as ethnic, nutritional, and environmental influences were not assessed, which could affect the interpretation of the hematological abnormalities observed.
CONCLUSION
This study highlights a high prevalence of hematological disturbances in pSLE, with anemia being the most common. The significant negative association between hemoglobin levels and disease activity status was a valuable clinical marker. Therefore, integrating routine hematological screening, with particular attention to hemoglobin trends, is recommended for effectively assessing disease activity status in pSLE patients.
AUTHORS’ CONTRIBUTION
The authors confirm their contribution to the paper as follows: K.L.: Methodology; M.A., M.I.I, S.A.R: Draft manuscript. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| SLE | = Systemic Lupus Erythematosus |
| pSLE | = Pediatric Systemic Lupus Erythematosus |
| AIHA | = Autoimmune hemolytic anemia |
| SLEDAI | = Systemic Lupus Erythematosus Disease Activity Index |
| ANA | = Anti-nuclear antibody |
| IF | = Immunofluorescence |
| UTP | = Urinary total protein |
ETHICAL APPROVAL AND CONSENT TO PARTICIPATE
Ethical clearance was taken from the Institutional Review Board of BSMMU (NO. BSMMU/2021/8748) before enrollment.
HUMAN AND ANIMAL RIGHTS
All procedures performed in studies involving human participants were in accordance with the ethical standards of institutional and/or research committee and with the 1975 Declaration of Helsinki, as revised in 2013.
AVAILABILITY OF DATA AND MATERIALS
All data generated or analyzed during this study are included in this published article.
ACKNOWLEDGEMENTS
Declared none.


